Posts Tagged Randomized controlled trial
[Abstract] Effects of Circuit Class Training Versus Individual, Task Specific Training on Upper Extremity Function in Chronic Stroke Patients
Posted by Kostas Pantremenos in Paretic Hand, Spasticity on February 15, 2024
Abstract

Background: Stroke is a leading contributor to disability globally, emphasizing the need for effective rehabilitation techniques. Circuit class training (CCT) and individual, task-specific training (ITST) have emerged as potential approaches for enhancing upper extremity function in stroke survivors. Comparative analyses of their efficacy, especially among chronic stroke patients, are scant.
Objective: This study aimed to evaluate and compare the impacts of CCT and ITST on upper extremity spasticity, motor function, and quality of life in individuals with chronic stroke.
Methods: In a randomized controlled trial, 36 chronic stroke patients were allocated to either CCT or ITST groups. Participants were aged 45-70 years, had experienced a single stroke episode, and were at least 6 months post-stroke, with specific inclusion criteria regarding spasticity and motor function levels. The interventions were delivered for 1.5 hours daily, five days a week, over eight weeks. Outcomes were measured using the Modified Ashworth Scale (MAS) for spasticity, Functional Independence Measure for Upper Extremity (FMA-UE) for motor function, and Stroke-Specific Quality of Life (SS-QOL) scale for quality of life, analyzed using SPSS version 25.
Results: Post-intervention, both CCT and ITST participants exhibited significant improvements in their outcomes. MAS scores showed a reduction in spasticity, with average improvements not significantly differing between the groups. FMA-UE scores increased by an average of 10 points in both groups, indicating enhanced motor function without a significant difference between the groups (p > 0.05). SS-QOL scores improved by an average of 20 points in each group, reflecting better quality of life, with no significant intergroup difference observed.
Conclusion: The study concludes that CCT and ITST are equally effective in ameliorating upper extremity spasticity, motor function, and quality of life among chronic stroke patients. The selection between CCT and ITST can thus be personalized based on patient preferences, available resources, and logistical considerations, maintaining rehabilitation efficacy.
References
Adeagbo CA, Olawale OA, Gbiri CAO. Transcranial direct current stimulation and repetitive functional task-oriented programme for upper limb functional rehabilitation in stroke survivors. Physical Therapy Reviews. 2021;26(6):420-7.
Alsubiheen AM, Choi W, Yu W, Lee H. The effect of task-oriented activities training on upper-limb function, daily activities, and quality of life in chronic stroke patients: A randomized controlled trial. International journal of environmental research and public health. 2022;19(21):14125.
Alwhaibi RM, Mahmoud NF, Zakaria HM, Badawy WM, Elzanaty MY, Ragab WM, et al. A comparative study on the effect of task specific training on right versus left chronic stroke patients. International journal of environmental research and public health. 2020;17(21):7950.
Anandan D, PK TN, Arun B, Priya V. Effect of task specific training with proprioceptive neuromuscular facilitation on stroke survivors. Biomedicine. 2020;40(3):363-6.
Bastola P, Singh P, Pinto D. A comparison of the effect of resistance training on upper extremity motor function, motor recovery, and quality of life in sub-acute stroke participants. Medical Journal of Dr DY Patil University. 2021;14(2):219-25.
Bovonsunthonchai S, Aung N, Hiengkaew V, Tretriluxana J. A randomized controlled trial of motor imagery combined with structured progressive circuit class therapy on gait in stroke survivors. Scientific Reports. 2020;10(1):6945.
da Silva ESM, Ocamoto GN, Santos-Maia GLd, de Fatima Carreira Moreira Padovez R, Trevisan C, de Noronha MA, et al. The effect of priming on outcomes of task-oriented training for the upper extremity in chronic stroke: a systematic review and meta-analysis. Neurorehabilitation and Neural Repair. 2020;34(6):479-504.
Deshpande S, Mohapatra S, Girish N. Influence of task-oriented circuit training on upper limb function among rural community-dwelling survivors of stroke. International Journal of Therapy And Rehabilitation. 2020;27(8):1-8.
Doğan M, Ayvat E, Kılınç M. Telerehabilitation versus virtual reality supported task-oriented circuit therapy on upper limbs and trunk functions in patients with multiple sclerosis: A randomized controlled study. Multiple Sclerosis and Related Disorders. 2023;71:104558.
Donnellan-Fernandez K, Ioakim A, Hordacre B. Revisiting dose and intensity of training: Opportunities to enhance recovery following stroke. Journal of Stroke and Cerebrovascular Diseases. 2022;31(11):106789.
Dorsch S, Carling C, Cao Z, Fanayan E, Graham PL, McCluskey A, et al. Bobath therapy is inferior to task-specific training and not superior to other interventions in improving arm activity and arm strength outcomes after stroke: a systematic review. Journal of physiotherapy. 2023;69(1):15-22.
Eldemir S, Guclu-Gunduz A, Eldemir K, Saygili F, Yilmaz R, Akbostancı MC. The effect of task-oriented circuit training-based telerehabilitation on upper extremity motor functions in patients with Parkinson’s disease: A randomized controlled trial. Parkinsonism & Related Disorders. 2023;109:105334.
Friel KM, Ferre CL, Brandao M, Kuo H-C, Chin K, Hung Y-C, et al. Improvements in upper extremity function following intensive training are independent of corticospinal tract organization in children with unilateral spastic cerebral palsy: a clinical randomized trial. Frontiers in Neurology. 2021;12:660780.
Gnanaprakasam A, Karthikbabu S, Ravishankar N, Solomon JM. Effect of task-based bilateral arm training on upper limb recovery after stroke: A systematic review and meta-analysis. Journal of Stroke and Cerebrovascular Diseases. 2023;32(7):107131.
Hsu H-Y, Kuan T-S, Tsai C-L, Wu P-T, Kuo Y-L, Su F-C, et al. Effect of a novel perturbation-based pinch task training on sensorimotor performance of upper extremity for patients with chronic stroke: A pilot randomized controlled trial. Archives of Physical Medicine and Rehabilitation. 2021;102(5):811-8.
Ibrahim R, Lawal IU, Joseph C. Intensity of Task-Specific Training for Functional Ability Post-stroke: Protocol for a Systematic Review. 2021.
Johar MN, Nordin NAM, Yusoff YAM. Effects of Game-Based Task-Oriented Circuit Training on Physical Functions of Stroke Survivors: A Pilot Study in A State Hospital in Kelantan, Malaysia. Asian Journal of Medicine and Biomedicine. 2021;5(S2):5-14.
Kim S-M, Kang S-H. The Effects of Task-Oriented Circuit Training Using Unstable Surface on Balance, Walking and Balance Confidence in Subacute Stroke Patients. Journal of The Korean Society of Integrative Medicine. 2021;9(4):211-23.
Lima ACd, Christofoletti G. Exercises with action observation contribute to upper limb recovery in chronic stroke patients: a controlled clinical trial. Motriz: Revista de Educação Física. 2020;26.
Llorens R, Fuentes MA, Borrego A, Latorre J, Alcañiz M, Colomer C, et al. Effectiveness of a combined transcranial direct current stimulation and virtual reality-based intervention on upper limb function in chronic individuals post-stroke with persistent severe hemiparesis: A randomized controlled trial. Journal of neuroengineering and rehabilitation. 2021;18:1-13.
Lotter JK, Henderson CE, Plawecki A, Holthus ME, Lucas EH, Ardestani MM, et al. Task-specific versus impairment-based training on locomotor performance in individuals with chronic spinal cord injury: a randomized crossover study. Neurorehabilitation and neural repair. 2020;34(7):627-39.
Martins JC, Nadeau S, Aguiar LT, Scianni AA, Teixeira-Salmela LF, De Morais Faria CDC. Efficacy of task-specific circuit training on physical activity levels and mobility of stroke patients: A randomized controlled trial. NeuroRehabilitation. 2020;47(4):451-62.
Mattos DJ, Rutlin J, Hong X, Zinn K, Shimony JS, Carter AR. White matter integrity of contralesional and transcallosal tracts may predict response to upper limb task-specific training in chronic stroke. NeuroImage: Clinical. 2021;31:102710.
Mawase F, Cherry-Allen K, Xu J, Anaya M, Uehara S, Celnik P. Pushing the rehabilitation boundaries: hand motor impairment can be reduced in chronic stroke. Neurorehabilitation and neural repair. 2020;34(8):733-45.
McDonell I, Barr C, van den Berg M. Implementing circuit class training can increase therapy time and functional independence in people with stroke receiving inpatient rehabilitation: findings from a retrospective observational clinical audit. Physiotherapy Theory and Practice. 2023:1-7.
Mooney RA, Cirillo J, Stinear CM, Byblow WD. Neurophysiology of motor skill learning in chronic stroke. Clinical Neurophysiology. 2020;131(4):791-8.
Nath D, Singh N, Saini M, Banduni O, Kumar N, Srivastava MP, et al. Clinical potential and neuroplastic effect of targeted virtual reality based intervention for distal upper limb in post-stroke rehabilitation: a pilot observational study. Disability and Rehabilitation. 2023:1-10.
Palimeris S. Combining a Tailored Strength Training Program with Transcranial Direct-Current Stimulation (tDcs) to Improve Upper Extremity Function in Chronic Stroke Patients: McGill University (Canada); 2020.
Roos MA, Thielman GT, Packel L, Moelter ST, Khakhina S, Klase ZA. The Impact of a Functional Circuit Training Program in People with ChronicStroke: A Non-Randomized Feasibility Study. 2021.
Zaman T, Mukhtar T, Waseem Zaman M, Shahid MN, Bibi S, Fatima A. Effects of task-oriented training on dexterous movements of hands in post stroke patients. International Journal of Neuroscience. 2024;134(2):175-83.
[ARTICLE] Can specific virtual reality combined with conventional rehabilitation improve poststroke hand motor function? A randomized clinical trial – Full Text
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION, Virtual reality rehabilitation on April 8, 2023
Abstract
Trial objective
To verify whether conventional rehabilitation combined with specific virtual reality is more effective than conventional therapy alone in restoring hand motor function and muscle tone after stroke.
Trial design
This prospective single-blind randomized controlled trial compared conventional rehabilitation based on physiotherapy and occupational therapy (control group) with the combination of conventional rehabilitation and specific virtual reality technology (experimental group). Participants were allocated to these groups in a ratio of 1:1. The conventional rehabilitation therapists were blinded to the study, but neither the participants nor the therapist who applied the virtual reality–based therapy could be blinded to the intervention.
Participants
Forty-six patients (43 of whom completed the intervention period and follow-up evaluation) were recruited from the Neurology and Rehabilitation units of the Hospital General Universitario of Talavera de la Reina, Spain.
Intervention
Each participant completed 15 treatment sessions lasting 150 min/session; the sessions took place five consecutive days/week over the course of three weeks. The experimental group received conventional upper-limb strength and motor training (100 min/session) combined with specific virtual reality technology devices (50 min/session); the control group received only conventional training (150 min/session).
Results
As measured by the Ashworth Scale, a decrease in wrist muscle tone was observed in both groups (control and experimental), with a notably larger decrease in the experimental group (baseline mean/postintervention mean: 1.22/0.39; difference between baseline and follow-up: 0.78; 95% confidence interval: 0.38–1.18; effect size = 0.206). Fugl-Meyer Assessment scores were observed to increase in both groups, with a notably larger increase in the experimental group (total motor function: effect size = 0.300; mean: − 35.5; 95% confidence interval: − 38.9 to − 32.0; wrist: effect size = 0.290; mean: − 5.6; 95% confidence interval: − 6.4 to − 4.8; hand: effect size = 0.299; mean: − -8.9; 95% confidence interval: − 10.1 to − 7.6). On the Action Research Arm Test, the experimental group quadrupled its score after the combined intervention (effect size = 0.321; mean: − 32.8; 95% confidence interval: − 40.1 to − 25.5).
Conclusion
The outcomes of the study suggest that conventional rehabilitation combined with a specific virtual reality technology system can be more effective than conventional programs alone in improving hand motor function and voluntary movement and in normalizing muscle tone in subacute stroke patients. With combined treatment, hand and wrist functionality and motion increase; resistance to movement (spasticity) decreases and remains at a reduced level.[…]

[ARTICLE] Virtual Reality Training Using Nintendo Wii Games for Patients With Stroke: Randomized Controlled Trial – Full Text
Posted by Kostas Pantremenos in Paretic Hand, Video Games/Exergames, Virtual reality rehabilitation on December 12, 2022
Abstract
Background
Stroke is a leading cause of disability. It is difficult to devise an optimal rehabilitation plan once stroke survivors are back home. Conventional rehabilitative therapies are extensively used in patients with stroke to recover motor functioning and disability, but these are arduous and expensive. Virtual reality (VR) video games inspire patients to get involved in their therapeutic exercise routine in a fun way. VR in the form of games provides a fruitful, secure, and challenging learning environment for motor control and neural plasticity development in rehabilitation. The effects of upper limb sensorimotor functioning and balance are the main focus of this trial.
Objective
The aim of this study is to compare the effects of VR training and routine physical therapy on balance and upper extremity sensorimotor function in patients with stroke.
Methods
It was a single assessor-blinded randomized clinical trial. A total of 74 participants with their first chronic stroke were included and rehabilitated in a clinical setting. The lottery method was used to randomly assign patients to either the VR group (n=37) or the routine physical therapy group (n=37). The VR group received a 1-hour session of VR training for 3 weekdays over 6 weeks, and the routine physical therapy group received different stretching and strengthening exercises. The outcome measuring tools were the Berg Balance Scale for balance and the Fugl-Meyer Assessment (upper extremity) scale for sensorimotor, joint pain, and range assessment. The assessment was done at the start of treatment and after the 6 weeks of intervention. Data analysis was done using SPSS 22.
Results
The trial was completed by 68 patients. A significant difference between the two groups was found in the Berg Balance Scale score (P<.001), Fugl-Meyer Assessment for motor function (P=.03), and Fugl-Meyer Assessment for joint pain and joint range (P<.001); however, no significant difference (P=.19) in the Fugl-Meyer Assessment for upper extremity sensation was noted.
Conclusions
VR training is helpful for improving balance and function of the upper extremities in the routine life of patients with stroke; although, it was not found to be better than conventional training in improving upper limb sensation. VR training can be a better option in a rehabilitation plan designed to increase functional capability.
Introduction
Stroke is a leading cause of disability, and it is difficult to devise an optimal rehabilitation plan for patients with stroke once they are discharged from the hospital [1]. Almost 85% of patients with stroke have hemiparesis after stroke while 55% to 75% of stroke survivors have motor dysfunction. South Asian people (people of India, Pakistan, Sri Lanka, Bangladesh, Nepal, and Bhutan) have a higher risk of stroke because of compromised cardiac and metabolic rate [2,3]. Treatment for stroke is initiated with drugs [4], and surgery might be another option to repair any constriction or narrowing of blood vessels [5,6]. Patient rehabilitation is an important part of treatment. The main purpose of rehabilitation is to improve the quality of life for patients with stroke and make them independent [7,8]. Conventional rehabilitative therapies are extensively used to help patients with stroke recover motor functioning and disability. However, the application of conventional techniques is arduous and expensive, and requires transportation of patients to tertiary care hospitals especially in countries like Pakistan where hospitals are less in number. Virtual reality (VR) training in the form of games [9] provides a fruitful, secure, and challenging learning environment for motor control and neural plasticity development after stroke. VR video games inspire patients to get involved in their therapeutic exercise routine in a fun way [10]. Depending on the remodeling and reorganization of brain function, previous studies found that VR can be a great alternative for quick functional recovery after stroke [11]. Mirror neurons in the cortex can be activated through observational learning by VR training. Participants who received sensory input in VR training were also more likely to learn the desired motor behavior [12]. The feedback can help to promote the development of use-dependent cortical plasticity, which could lead to improved motor control. Furthermore, the functional improvement induced by VR training could significantly boost participants’ confidence and self-efficacy in a new environment. Moreover, another advantage of VR is that it can greatly save on labor and cost of patients [13].
Stroke is seen to be more prevalent in countries like Pakistan, as the people are more inclined toward using local drugs like naswar, pipe smoking, and beetle leaf chewing (paan). Thus, there is higher incidence of stroke in middle-aged populations (<45 years) [14–16]. The study aimed for a younger population with stroke and found cost-effective treatment protocols at the same time. The unique needs of young people with stroke and the promising opportunity provided by a low-cost serious game would be a beneficial addition in treatment strategy. There is inadequate evidence in the literature to generalize effects on upper limb sensorimotor function and gait through commercial gaming in young patients with stroke. Studies on effectiveness of VR programs in comparison to traditional methods on functional-motor improvement of an upper limb are needed in low-resource countries to reduce cost and time through target-oriented interventions. This study was conducted to compare the effects of VR training and routine physical therapy on balance and function of upper limbs in patients with stroke from the lower- and middle-class populations. This study is conducted to accept or reject the hypothesis that VR has a significantly better effect on balance and upper limb function in patients with stroke.[…]
[Abstract] Effects of neurological music therapy on behavioural and emotional recovery after traumatic brain injury: A randomized controlled cross-over trial
Posted by Kostas Pantremenos in Cognitive Rehabilitation, Music/Music therapy, TBI on September 21, 2022
Abstract
Traumatic brain injury (TBI) causes deficits in executive function (EF), as well as problems in behavioural and emotional self-regulation. Neurological music therapy may aid these aspects of recovery. We performed a cross-over randomized controlled trial where 40 persons with moderate-severe TBI received a 3-month neurological music therapy intervention (2 times/week, 60 min/session), either during the first (AB, n = 20) or second (BA, n = 20) half of a 6-month follow-up period. The evidence from this RCT previously demonstrated that music therapy enhanced general EF and set shifting. In the current study, outcome was assessed with self-report and caregiver-report questionnaires performed at baseline, 3-month, 6-month, and 18-month stages. The results showed that the self-reported Behavioural Regulation Index of the Behaviour Rating Inventory of Executive Function (BRIEF-A) improved more in the AB than BA group from baseline to 3-month stage and the effect was maintained in the 6-month follow-up. No changes in mood or quality of life questionnaires were observed. However, a qualitative content analysis of the feedback revealed that many participants experienced the intervention as helpful in terms of emotional well-being and activity. Our results suggest that music therapy has a positive effect on everyday behavioural regulation skills after TBI.
Similar articles
- Neurological Music Therapy Rebuilds Structural Connectome after Traumatic Brain Injury: Secondary Analysis from a Randomized Controlled Trial.Sihvonen AJ, Siponkoski ST, Martínez-Molina N, Laitinen S, Holma M, Ahlfors M, Kuusela L, Pekkola J, Koskinen S, Särkämö T.J Clin Med. 2022 Apr 14;11(8):2184. doi: 10.3390/jcm11082184.PMID: 35456277 Free PMC article.
- Music Therapy Enhances Executive Functions and Prefrontal Structural Neuroplasticity after Traumatic Brain Injury: Evidence from a Randomized Controlled Trial.Siponkoski ST, Martínez-Molina N, Kuusela L, Laitinen S, Holma M, Ahlfors M, Jordan-Kilkki P, Ala-Kauhaluoma K, Melkas S, Pekkola J, Rodriguez-Fornells A, Laine M, Ylinen A, Rantanen P, Koskinen S, Lipsanen J, Särkämö T.J Neurotrauma. 2020 Feb 15;37(4):618-634. doi: 10.1089/neu.2019.6413. Epub 2019 Dec 5.PMID: 31642408 Clinical Trial.
- Cognitive efficacy and neural mechanisms of music-based neurological rehabilitation for traumatic brain injury.Martínez-Molina N, Siponkoski ST, Särkämö T.Ann N Y Acad Sci. 2022 Sep;1515(1):20-32. doi: 10.1111/nyas.14800. Epub 2022 Jun 8.PMID: 35676218 Review.
- A group memory rehabilitation programme for people with traumatic brain injuries: the ReMemBrIn RCT.das Nair R, Bradshaw LE, Carpenter H, Clarke S, Day F, Drummond A, Fitzsimmons D, Harris S, Montgomery AA, Newby G, Sackley C, Lincoln NB.Health Technol Assess. 2019 Apr;23(16):1-194. doi: 10.3310/hta23160.PMID: 31032782 Free PMC article. Clinical Trial.
- Evaluation of the short-term executive plus intervention for executive dysfunction after traumatic brain injury: a randomized controlled trial with minimization.Cantor J, Ashman T, Dams-O’Connor K, Dijkers MP, Gordon W, Spielman L, Tsaousides T, Allen H, Nguyen M, Oswald J.Arch Phys Med Rehabil. 2014 Jan;95(1):1-9.e3. doi: 10.1016/j.apmr.2013.08.005. Epub 2013 Aug 27.PMID: 23988395 Clinical Trial.
[ARTICLE] Effectiveness of robot-assisted virtual reality mirror therapy for upper limb motor dysfunction after stroke: study protocol for a single-center randomized controlled clinical trial – Full Text
Posted by Kostas Pantremenos in Mirror therapy, Rehabilitation robotics, Virtual reality rehabilitation on September 5, 2022
Abstract
Background
Upper limb motor dysfunction is a common sequela of stroke, and its clinical efficacy needs to be improved. This protocol describes a trial to verify the clinical efficacy of robot-assisted virtual reality mirror therapy (RAVRMT) in improving upper limb motor dysfunction in stroke patients, and to explore the central mechanism by using functional magnetic resonance imaging (fMRI).
Methods
This trial will be a single-center, assessor-blinded, randomized controlled clinical study. Thirty-two eligible patients will be randomly divided into 2 groups according to the ratio of 1:1, namely virtual reality mirror therapy (VRMT) group and robot-assisted virtual reality mirror therapy (RAVRMT) group. The interventions will be performed once a day for 4 weeks. Primary outcome is Fugl–Meyer motor function assessment-Upper Extremity (FMA-UE), secondary outcomes are the Montreal Cognitive Assessment (MoCA), activities of daily living (ADL), quality of life (QOL), the pain visual analogue scale (VAS-pain) and fMRI. Adverse events will be recorded, and severe adverse events will be used as criteria to discontinue the intervention.
Discussion
Combined application of robot-assisted therapy and virtual reality mirror therapy could theoretically activate mirror neuron system and reward circuits to a greater extent, but further high-quality research is needed. The results of this trial will determine whether RAVRMT could better improve upper limb motor dysfunction after stroke and explore its central mechanism using fMRI.
Background
In recent years, stroke has become the second leading cause of death and the third leading cause of disability in the world [1]. Studies have shown that about 80% of acute stroke patients and more than 55% chronic stroke patients have upper limb motor dysfunction [2, 3], which seriously affects the quality of life of patients and their families [4, 5]. At present, the conventional rehabilitation methods for upper limb motor dysfunction after stroke consists of physical therapy [6], occupational therapy [7], but more than 50% of stroke patients still have long-term upper limb motor dysfunction [8]. The clinical efficacy of conventional rehabilitation methods in the treatment of upper limb motor dysfunction after stroke is not satisfactory, and new rehabilitation concepts and techniques are urgently needed.
How can we better promote the recovery of upper limb motor dysfunction after stroke? The disruption of motor-related brain networks is the primary cause of upper limb motor dysfunction after stroke, and another important cause is the upper limb disuse caused by long-term inactivity, so a single treatment for one cause is not adequate [9]. In stroke, the disruption of motor-related brain networks is the damage at the central level [10] and the upper limb disuse caused by long-term inactivity is the damage at the peripheral level [11]. As a clinical central intervention technology, mirror therapy can activate the motor-related brain network through action observation [12]. A recent meta-analysis demonstrates that mirror therapy can significantly improve upper limb motor dysfunction in stroke patients [13]. As a new kind of mirror therapy, virtual reality mirror therapy (VRMT) can provide immersion mirror therapy for stroke patients [14]. However, mirror therapy lacks the participation of proprioception [15], which may not be able to simulate the actual scene well and activate the motor-related brain network to the greatest extent. As a clinical peripheral intervention technology, a large number of studies have proved that robot-assisted therapy (RAT) can have a positive impact on the recovery of upper limb motor dysfunction in stroke patients through programmed task training [16, 17], including motor function and proprioception. At present, more and more studies show that mirror therapy combined with other interventions is superior to a single rehabilitation therapy in the treatment of upper limb motor dysfunction in stroke patients [18–20].
In this study, we designed a device that combined RAT and immersion VRMT to treat upper limb motor dysfunction after stroke. Moreover, the device can dynamically match the motion trajectory of the RAT with the motion images displayed in real time by the VRMT, so as to realize the cycle of “visual information input-visual information output-motion feedback” in the stroke patient. This is different from other combinations of a central intervention and a peripheral intervention for upper limb motor dysfunction after stroke [21, 22], which a central intervention and a peripheral intervention do not dynamically match each other in real time. In summary, the device is an innovative medical rehabilitation machine, which realizes the cycle of “visual information input-visual information output-motion feedback”, and provides patients with a full-body immersive experience that is consistent with vision, touch, and proprioception. We call the treatment robot-assisted virtual reality mirror therapy (RAVRMT) and the device RAVRMT robot. The RAVRMT robot is ultimately expected to activate mirror neuron system (MNS) and reward circuits to the greatest extent possible, thereby promoting the improvement of upper limb motor dysfunction in patients after stroke.
Therefore, we designed a randomized controlled trial to explore the clinical efficacy of RAVRMT in improving upper limb motor dysfunction in patients after stroke. Meanwhile, in this trial, functional magnetic resonance imaging (fMRI) scans will be performed to find significant changes in motor-related brain functional connectivity, and the motor-related brain functional network will be compared before and after the intervention to further reveal the brain neural mechanism of RAVRMT’s effects.[…]

Schematic diagram of virtual reality mirror therapy
[Abstract] IMproving Physical ACtivity after stroke via Treadmill training (IMPACT) and self-management: A randomized trial
Posted by Kostas Pantremenos in Gait Rehabilitation - Foot Drop on July 15, 2022
Abstract
Aim:
To determine if treadmill training embedded in self-management education commencing during stroke inpatient rehabilitation results in more physical activity than usual gait training.
Method:
A prospective, parallel-group, randomized trial with concealed allocation, blinded measurement, and intention-to-treat analysis involving 119 stroke survivors undergoing rehabilitation who were able to walk independently was undertaken. The experimental group undertook treadmill training (40–60% heart rate reserve) and self-management education for 30 min, three times a week for 8 weeks, and the control group undertook the same amount of usual gait training. Outcomes were measured at baseline (Week 0), on completion of the intervention (Week 8), and beyond the intervention (Week 26). The primary outcome was physical activity measured as steps/day using an activity monitor. Secondary outcomes were walking ability, cardiorespiratory fitness, cardiovascular risk, depression, self-efficacy, perception of physical activity, participation, and quality of life.
Results:
After 8 weeks, the experimental group took 1436 more steps/day (95% confidence interval (CI) = 229 to 2643) than the control group. By 6 months, they took 871 more steps/day (95% CI −385 to 2129) than the control group. There was no difference between groups in any other outcome.
Conclusion:
In individuals undergoing rehabilitation after stroke, 8 weeks of treadmill training embedded in self-management resulted in more physical activity than usual gait training and this was largely maintained at 6 months, despite little effect on walking or cardiorespiratory fitness, suggesting the self-management was responsible.
[ARTICLE] The movement-to-music (M2M) study: study protocol for a randomized controlled efficacy trial examining a rhythmic teleexercise intervention for people with physical disabilities – Full Text
Posted by Kostas Pantremenos in Music/Music therapy, Tele/Home Rehabilitation on November 15, 2021
Abstract
Background
People with physical disabilities need exercise routines that are enjoyable, readily available in the home, adapted to their functional level, and eliminate common barriers to exercise participation related to transportation and time commitment. The purpose of the movement-to-music (M2M) study is to address these issues by establishing a remotely delivered, rhythmic exercise program for people with physical disabilities.
Methods
The study is a two-arm randomized controlled efficacy trial examining a 12-week remotely delivered M2M intervention (eM2M) in 108 people with physical disabilities. The primary outcomes are changes in cardiorespiratory fitness and muscle strength at post 12-week intervention.
Discussion
The eM2M study will enhance our understanding of an alternative intervention design and delivery mode that removes common barriers to exercise participation experienced by people with physical disabilities. The eM2M intervention may be an alternative option for people with physical disabilities to obtain regular exercise, especially during a pandemic when exercising in indoor facilities may be problematic.
………
Introduction
Background and rationale {6a}
Exercise trials for people with physical disabilities are currently challenged by the inability to reach large, heterogenous groups of participants that are representative of the target population. Studies typically incorporate highly controlled designs and focus on examining one specific disability group in an exercise intervention with an average sample size of 40 participants per study [1, 2]. Because of this, the generalizability and transferability of the research is limited, which creates a need to identify evidence-based exercise interventions that are robust enough to be successfully used by multiple disability groups [3]. To address this limitation, one potential strategy is for studies to include people within the exercise intervention based upon their physical function, as opposed to a specific type of disability.
Creating exercise interventions for different levels of physical function present a few challenges. For example, when an exercise intervention is delivered to participants with various levels of functional mobility, people with higher or lower levels of physical function may perceive the program as not challenging or too difficult, which may compromise the physiological adaptations achieved from the intervention. Froehlich-Grobe et al. examined the effectiveness of a physical activity intervention for women with a wide variety of mobility limitations and reported no significant physiologic change [4]. They suggested that a possible reason for this nonsignificant finding was the variability in functional mobility levels. The authors recommended that in future studies, researchers should design exercise interventions using a functionally based approach to ensure that every participant receives a similar exercise stimulus.
In addition to addressing the physiologic training needs in people with physical disabilities, it is also important to ensure that the interventions are accessible. Common barriers to exercise experienced by people with physical disabilities include lack of transportation to and from exercise venues, cost of the program, inaccessible fitness facilities, and time constraints [5]. Fortunately, there are a growing number of scalable telehealth technologies that have the potential to reach large numbers of people with disabilities in the comfort of their home [6]. In a recent review, we found that technology-driven exercise intervention studies sustained 56% of all outcomes measured at follow-up, compared to 24% for the exercise intervention studies that used technology only for measurement purposes [7]. Given the low rate of exercise participation among people with physical disabilities [8], there is a pressing need to continue exploring the potential of using telehealth technologies to deliver home-based exercise, or what we refer to as teleexercise, among people with disabilities.
Providing exercise that is enjoyable is also important as it will likely increase adherence to the exercise program in people with physical disabilities. Many fitness-related training protocols such as treadmill walking, stationary cycling, and weight lifting often have low adherence rates due to lack of enjoyment or social interaction [9,10,11]. Asano et al. surveyed 417 people with multiple sclerosis and found “dislike exercise” and “find exercise boring” were common reasons for not exercising [12]. Providing alternative and enjoyable forms of exercise thus becomes critical in helping individuals find activities that fit their interests and needs. Several studies have reported that music is an effective strategy for improving exercise adherence [13,14,15,16] and enjoyment [17, 18]. Listening to music during exercise is associated with positive moods and feelings [19]. Indeed, evidence suggests that dance is a promising intervention for improving physical function in people with Parkinson’s [20, 21], stroke [22], and other disabling conditions [23,24,25]. An additional benefit is that movement-based exercise with music can be performed almost anywhere with minimal equipment.[…]
[Abstract] Effects of neurological music therapy on behavioural and emotional recovery after traumatic brain injury: A randomized controlled cross-over trial
Posted by Kostas Pantremenos in Cognitive Rehabilitation, Music/Music therapy on September 10, 2021
Abstract
Traumatic brain injury (TBI) causes deficits in executive function (EF), as well as problems in behavioural and emotional self-regulation. Neurological music therapy may aid these aspects of recovery. We performed a cross-over randomized controlled trial where 40 persons with moderate-severe TBI received a 3-month neurological music therapy intervention (2 times/week, 60 min/session), either during the first (AB, n = 20) or second (BA, n = 20) half of a 6-month follow-up period. The evidence from this RCT previously demonstrated that music therapy enhanced general EF and set shifting. In the current study, outcome was assessed with self-report and caregiver-report questionnaires performed at baseline, 3-month, 6-month, and 18-month stages. The results showed that the self-reported Behavioural Regulation Index of the Behaviour Rating Inventory of Executive Function (BRIEF-A) improved more in the AB than BA group from baseline to 3-month stage and the effect was maintained in the 6-month follow-up. No changes in mood or quality of life questionnaires were observed. However, a qualitative content analysis of the feedback revealed that many participants experienced the intervention as helpful in terms of emotional well-being and activity. Our results suggest that music therapy has a positive effect on everyday behavioural regulation skills after TBI.
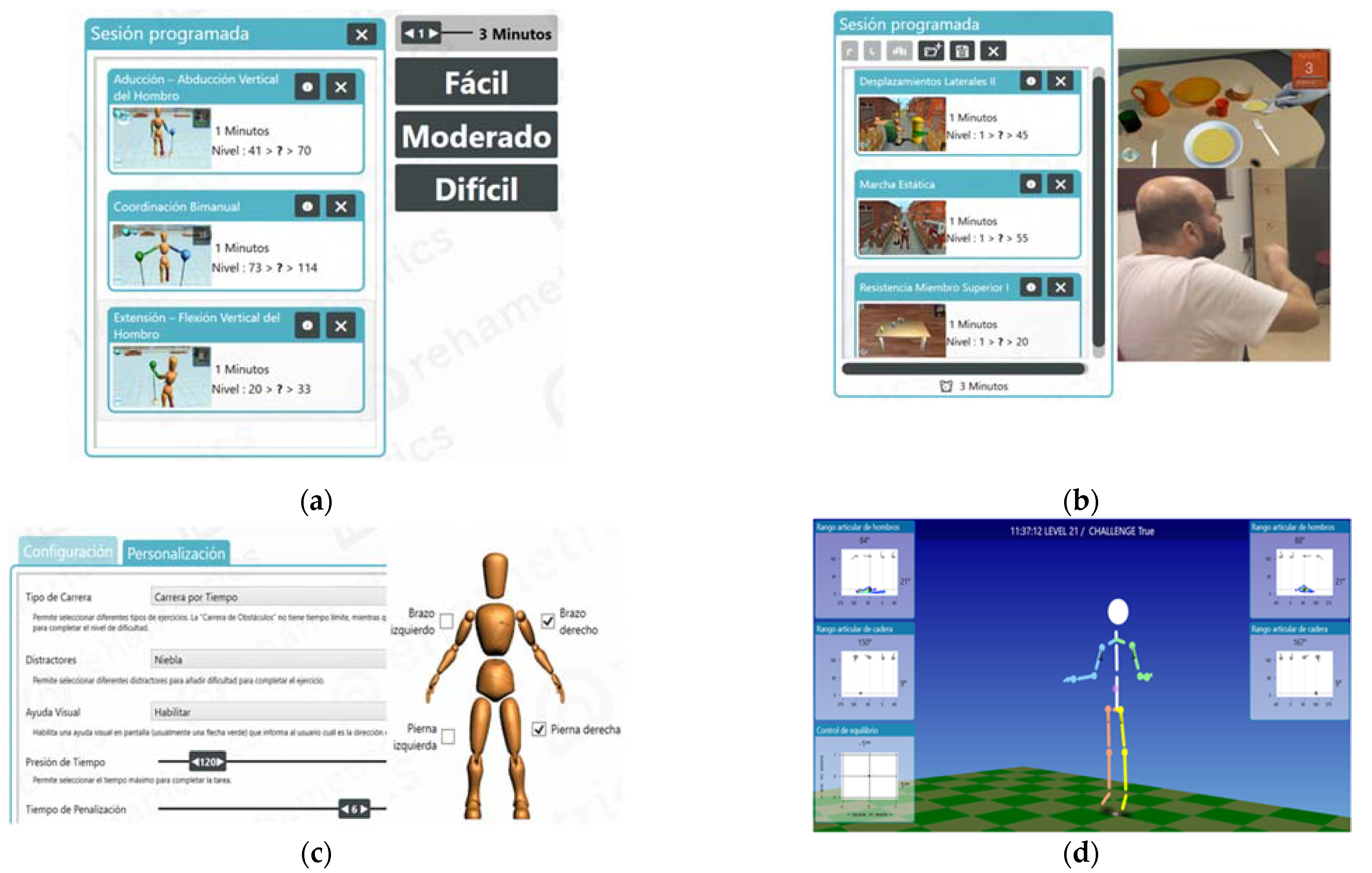
[ARTICLE] Effects of Specific Virtual Reality-Based Therapy for the Rehabilitation of the Upper Limb Motor Function Post-Ictus: Randomized Controlled Trial – Full Text
Posted by Kostas Pantremenos in Paretic Hand, Virtual reality rehabilitation on August 2, 2021
Abstract
This research analyzed the combined effect of conventional treatment and virtual reality exposure therapy on the motor function of the upper extremities in people with stroke. We designed a randomized controlled trial set in the rehabilitation and neurology departments of a hospital (Talavera de la Reina, Spain). The subjects included 43 participants, all randomized into experimental (conventional treatment + virtual reality exposure therapy) and control group (conventional treatment).; The main measures were Fugl-Meyer Assessment for upper extremity, Modified Ashworth Scale, and Stroke Impact Scale 3.0. The results included 23 patients in the experimental (62.6 ± 13.5 years) and 20 in the control group (63.6 ± 12.2 years) who completed the study. After the intervention, muscle tone diminished in both groups, more so in the experimental group (mean baseline/post-intervention: from 1.30 to 0.60; η2 = 0.237; p = 0.001). Difficulties in performing functional activities that implicate the upper limb also diminished. Regarding the global recovery from stroke, both groups improved scores, but the experimental group scored significantly higher than the controls (mean baseline/post-intervention: from 28.7 to 86.5; η2 = 0.633; p = 0.000). In conclusion, conventional rehabilitation combined with specific virtual reality seems to be more efficacious than conventional physiotherapy and occupational therapy alone in improving motor function of the upper extremities and the autonomy of survivors of stroke in activities of daily living.
1. Introduction
Stroke is one of the main causes of acquired disability in adulthood. The stroke epidemic is primarily driven by the aging of the world population, globalization and the urbanization of community settings [1,2]. The Stroke Alliance for Europe states that, every 20 s, a new case of stroke is detected in the adult population and predicts that the number of people affected will increase by 35% to 12 million people in 2040. As a result, it is estimated that the health and social costs for stroke diagnosis will increase to 75 million in 2030 (26% more than in 2017). In Spain, 550,941 people were diagnosed with stroke in 2017, generating a health expenditure of 1700 million euros and a total cost to the Spanish state of 3557 million euros [3].Around 80% of survivors present motor difficulties in the upper extremities, affecting the carrying out of activities of daily living (ADLs), the performance of roles in the community and the health-related quality of life (HRQoL) [4,5,6].Complications after stroke diagnosis can persist over time. Two-thirds of survivors are disabled 15 years later, two out of five are immersed in depressive states and more than a quarter develop cognitive impairment [7]. The costs derived from stroke diagnosis are high for survivors and their families, making their rehabilitation and survival processes a great challenge for health policymakers [8,9]. On average, an informal (non-professional) caregiver in Spain invests 2833 h per year in caring for the person affected by stroke and with limitations in ADLs [3].The general objective of neurological rehabilitation is to promote a rapid recovery from the multiple deficits after a stroke and the achievement of a lifestyle similar to the premorbid state [10,11]. Of all people diagnosed with stroke, only 30–40% regain certain skills in the upper limb after six months of intervention [12]. The upper limb remains non-functional for ADLs in up to 66% of survivors [13], constituting the most disabling of all residual disorders.In recent years, the use of neurorehabilitation approaches based on technology and virtual reality has increased, allowing the creation of effective rehabilitation environments and providing multimodal, controllable, and customizable stimulation [14], in which the recreation of virtual objects maximize visual feedback [15] and high intensity and high number of repetitions are key factors that influence neuroplasticity and functional improvement in patients [16]. Rehabilitation based on virtual reality offers the possibility of individualizing treatment needs, and at the same time, standardizing evaluation and training protocols [17,18]. In this sense, specific virtual reality technology for rehabilitation processes of people with neurological pathology allows working in a functional way and with specific intervention objectives, in addition to easily qualifying and documenting progress during the session [19]. Taking advantage of these characteristics, several researchers have used virtual reality exposure therapy (VRET) to recover motor function after stroke. In the treatment of the upper limb, studies indicate that this rehabilitation approach produces better motor and functional results than conventional therapy [20,21].The increasing clinical use of neurorehabilitation approaches based on technology and virtual reality leads to the assumption that spatial representations in virtual environments may vary slightly from the perceptions that the patient would experience in real spaces. In this sense, the team of Hruby et al. [22] insisted that spatial representations based in virtual reality systems should be realistic 1:1 replicas with regard to the individual characteristics of the subjects interacting with both virtual and real environments. This demand increases the validity of virtual reality techniques for therapeutic purposes, since interaction with a virtual space is safer and more profitable in the early phases of rehabilitation processes [23]. However, it is important for clinicians and researchers to consider that the interaction with a virtual environment continues to be different from the relationship that the subject maintains with the real environment [24] because people gradually build a mental representation of the geographic space that we work with or are immersed in. The locomotion techniques applied in the virtual model (software or hardware) can influence the cognitive representations of the person experiencing them [25].The present study aimed to analyze the combined effect of conventional treatment and VRET on motor function of the upper limb in people diagnosed with stroke in the acute phase and its evolution at three months in the Integrated Health Area of Talavera de la Reina.[…]