By Xavier Figueroa, Ph.D.
http://www.msktc.org/tbi/factsheets/Sexuality-After-Traumatic-Brain-Injury
 What is the largest sex organ in the body?
What is the largest sex organ in the body?
The brain, of course! (Followed by the spinal cord ganglia but let’s not judge).
Intimacy, desire, physical contact and pleasure, they are very basic needs in a relationship. Marriages, partnerships and friendships rely on this most basic link. But when a brain injury occurs, changes in desire and drive (hypo- and hyper-sexuality) can become apparent. Energy and mood can also be affected, which can induce a change in libido, interest and desire. Damage to certain portions of the brain may affect your ability to move, reducing spontaneity and self-esteem. Elements of coming to terms with the trauma, such as shock and recovery may take time, as well as recovery from physical rehabilitation. If the injury is chronic, other changes may become apparent, including cognitive and behavioral changes that shift how friends and partner interact with each other.
Much of these changes can occur days, weeks or even months after the injury, even in mild to moderate traumatic brain injuries. Knowing how to identify, adapt and overcome the changes associated with an ABI/TBI is an important part of recovery.
The most important information to take away from this post is the following: you are not alone, you are not abnormal and you will get better. Millions of individuals and couples have gone through the recovery of a brain injury and difficulties with reestablishing a functional sexual relationship. Hang in there.
What the Problem Looks Like
When we talk about sex, we are talking about something that is simple in practice, but complex in execution. Prior to the brain injury, a pattern of behavior between yourself and your partner was established. How you interacted and what you expected prior to and leading to sexual intimacy were established and anticipated. I wouldn’t call it a pattern (that’s not very exciting!), but a role in which you knew which part each one would play.
A brain injury directly affects the biggest and most important sex organ in the human body. It’s no wonder that sexual issues appear in 50-60% of people that suffer a moderate to severe TBI. In a recent article in US News and World Report (Health Day, April 29, 2013; Link) that reported on the study that appeared in NeuroRehabilitation: An International Journal:
‘The study found that 50 percent to 60 percent of people with TBI have sexual difficulties, such as reduced interest in sex, erectile dysfunction, pain during sex, difficulties in vaginal lubrication, difficulties achieving orgasm or staying aroused, and a sense of diminished sex appeal, Moreno said.
The research found that partners of those with TBI experienced personality and emotional changes, and a modification of family roles that can lead to a crisis, Moreno said. “For the spouse, the survivor becomes a different person, a person they do not recognize as the one they fell in love with in the past,” he said. “The spouse becomes a caregiver and this imbalance in the relationship directly affects sexual desire.”’
Even in cases of mild TBI, there are incidences of 25-50% of people experiencing sexual difficulties [1], especially in individuals exposed to bomb-blast injuries. Brain injuries are not mild…they can take a life of their own and totally transform who you are and how you relate to your significant other (spouse, partner or lover). Many of these changes can be divided into 5 major groups:
- Decreased Desire (Hyposexuality): inability to become interested in sex.
- Increased Desire (Hypersexuality): inappropriate sexual behavior; constant focus on sex.
- Decreased Arousal: Difficulty in achieving erection/lubrication.
- Difficulty or Inability to Reach Orgasm/Climax:
- Reproductive Changes: Low sperm count; missed periods.
But these are just the changes that occur with sexual interaction (as if that weren’t enough). These are behavioral changes that hide deeper and more profound changes that can occur throughout the body. Changes in sexual desire are like the proverbial canary in the coal mine…it warns you that something is amiss.
That Voodoo That You do…
Damage to the brain can induce a number of changes:
Fatigue/Tiredness
Hormonal Changes
Emotional Changes
Cognitive Changes
Spasticity/Movement Problems
These changes can come from very specific damage to certain areas of the brain, such as your pituitary, the frontal and temporal lobes of the brain. When you get down to it, sex is a very complicated process…neurologically speaking! A number of body systems have to work together to make the engines of desire go vroom…and when one system is not working, then it can cause the engine to misfire and stall.
The Tiny Organ
The pituitary gland is a tiny portion of the brain… but don’t let its size fool you. It is a master regulator of hormones that, when damaged, can diminish your ability to regulate your blood pressure, sleep cycle and hormones.
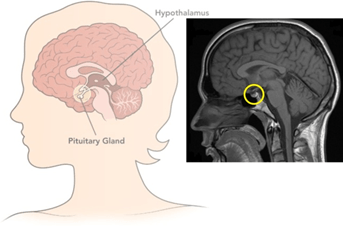 The function of the pituitary is diverse, as it can affect a number of really important functions:
The function of the pituitary is diverse, as it can affect a number of really important functions:
Hormones secreted from the pituitary gland help control the following body processes:
- Growth
- Blood pressure
- Pregnancy and stimulation of uterine contractions during childbirth
- Breast milk production
- Sex organ functions in both males and females
- Thyroid gland function
- The conversion of food into energy (metabolism)
- Water and osmolarity regulation in the body (which affects blood pressure)
- Water balance via the control of re-absorption of water by the kidneys
- Temperature regulation
- Pain relief
If that weren’t enough, this can cascade into disease states that may not seem related to a TBI. One thing that we are seeing with returning veterans is pituitary dysfunction is present and undiagnosed or under diagnosed. Even with hormone or growth factor replacement therapies, a pituitary that is not firing on all cylinders will continue to cause long-term problems. Although changes in sexual interaction are the most visible and can be due to pituitary damage, they warn that the damage is more profound. The Big Organ (the brain) has a lot of functions related to behavior…and when it comes to sex, behavior is key (good or bad).
The Tiny Brain (Hypothalamus)
This portion of the brain, the hypothalamus, is a close neighbor to the pituitary. So close, they are friends with benefits. One of the most important functions of the hypothalamus is to link the nervous system to the endocrine system via the pituitary gland (another name of the pituitary is the hypophysis).
The hypothalamus is more of a region than an actual structure. It is composed of many groups of neurons (called nuclei) that control a wide variety of hormonal secretions and behaviors. In a recent small scale study of severe TBI, it was discovered that ~21% of study subjects suffered from hypothalamic-hypophysial dysfunction. In about 40% of male TBI sufferers, there was a detectable drop in testosterone levels [2], which can affect sexual drive and desire in men. About 15% of all patients with a TBI have some degree of hypopituitarism that can go unrecognized and could be mistakenly ascribed to persistent neurologic injury and cognitive impairment [3].
The reason for the hypothalamic damage being mistaken for neurologic injury and cognitive impairment are due to the very broad effects that the hypothalamus exerts on metabolism and brain function. If the hypothalamus is misfiring, it takes a very involved physician (or physicians), with training in neurology, endocrinology and/or experience with TBI to identify the problem. A lot of systems can malfunction in a brain injury.
The Frontal Lobe

The frontal lobe (in green) • tumblr
In head injuries, damage to the frontal lobe is thought to occur frequently. Car crashes (especially front end collisions, are thought to cause frontal and occipital lobe damage. Damage to the frontal lobe has been reported to cause individuals to behave inappropriately in response to normal social situations. Loud or overly-boisterous exchanges, inappropriate genital touching (in public) or fixation on one subject or person have been reported outcomes after a TBI. Changes in emotional affect (expression of emotions) that are felt may not be expressed in the face or voice. For example, someone who is feeling happy would not smile, and his or her voice would be devoid of emotion. This can be very disconcerting to a partner and can be experienced a loss of affection or interest. How a partner or loved one that is a caretaker of a TBI victim experiences the injury will have a direct effect on their own sexual desire and interest.
Along the same lines, though, the person may also exhibit excessive, unwarranted displays of emotion or poor control of anger. Poor anger management is associated with some forms of frontal lobe damage. Depression is not an uncommon outcome from a head injury, especially if there is frontal lobe damage. Also common along with depression is a loss of or decrease in motivation. Someone might not want to carry out normal daily activities and would not feel “up to it”. Sex might not seem as interesting or motivating.
Those who are close to the person who has experienced the damage may notice that the person no longer behaves like him or herself. The frontal lobe is the same part of the brain that is responsible for executive functions such as planning for the future, judgment, decision-making skills, attention span, and inhibition. These functions can decrease drastically in someone whose frontal lobe is damaged. A short list of behavioral changes associated with frontal lobe damage is given below:
- Agitation
- Explosive anger and irritability
- Lack of awareness and insight
- Impulsivity and disinhibition
- Emotional lability
- Self-centeredness
- Apathy and poor motivation
- Depression
- Anxiety
- Inflexibility and obsessionality
- Sexual problems
Frontal lobe damage is only one part of cerebral cortex, but is the most common type of cortical damage due to a TBI. Other parts may be damaged as well. Frontal lobe damage is common and better associated with impulse and emotional control, making sufferers act completely out of character and unable to control or edit themselves or their responses.
Putting it Together
So, after reading all of this, what does it do for you? How does this help you re-establish the emotional, sexual and intimate relationship you wish with your partner? As a caretaker, or as a sufferer, the TBI is a big elephant in the room. It exists; it takes up space in your life, even though it can’t be seen. The person you knew is not present…they have not come back from their injury and they might not come back. Some do recover, others do not. But you can still create a new bond, a new relationship and a new life. And you can fight to repair the damage to the brain.
There are limited options for therapy in current medical practice. Mostly, it is focused on developing new skills, relearning old ones, developing coping skills or taking medications. That’s just for the TBI sufferer, not the caretaker(s). The complexity and variety of problems that pop-up when dealing with a brain injury are truly staggering and expensive. Fortunately, the majority of mild-to-moderate TBI’s do recover. Patience and persistence in therapy are required in order to make a recovery.
Unfortunately, for a portion of all TBI sufferers, recovery may take years. That is a long-time to wait. Therapies that help to re-build the brain connections (neuroplasticity) or restore blood flow to the brain hold promise for restoring function again. Hyperbaric oxygen therapy (HBOT) is one such therapy that has a good number of clinical studies to support its use for chronic TBI and PCS [4-9]. Near infra-red and infra-red technologies show promise for a TBI therapy, as well [10-13].
Nutritional support, such as Omega-3 fatty acids (DHA and EPA), has shown the ability to reduce the long-term neuroinflammation associated with a TBI [14-16] and help with white matter repair. Other nutritional therapies may exist to help mediate repair in a TBI.
The take home message is that there are potential therapies that are being developed to help treat the neurological damage of a TBI. Take heart that the “new normal” for yourself or your loved one may not need to be permanent.
- Wilkinson, C.W., et al., High prevalence of chronic pituitary and target-organ hormone abnormalities after blast-related mild traumatic brain injury. Front Neurol, 2012. 3: p. 11.
- Kopczak, A., et al., Screening for hypopituitarism in 509 patients with traumatic brain injury or subarachnoid hemorrhage. J Neurotrauma, 2014. 31(1): p. 99-107.
- Pekic, S. and V. Popovic, Chapter 18 – Alternative causes of hypopituitarism: traumatic brain injury, cranial irradiation, and infections, in Handbook of Clinical Neurology, M.K. Eric Fliers and A.R. Johannes, Editors. 2014, Elsevier. p. 271-290.
- Boussi-Gross, R., et al., Hyperbaric Oxygen Therapy Can Improve Post Concussion Syndrome Years after Mild Traumatic Brain Injury – Randomized Prospective Trial. PLoS One, 2013. 8(11): p. e79995.
- Wolf, G., et al., The effect of hyperbaric oxygen on symptoms after mild traumatic brain injury. J Neurotrauma, 2012. 29(17): p. 2606-12.
- Harch, P.G., et al., A phase I study of low-pressure hyperbaric oxygen therapy for blast-induced post-concussion syndrome and post-traumatic stress disorder. J Neurotrauma, 2012. 29(1): p. 168-85.
- Lin, J.W., et al., Effect of hyperbaric oxygen on patients with traumatic brain injury. Acta Neurochir Suppl, 2008. 101: p. 145-9.
- Shi, X.Y., et al., Evaluation of hyperbaric oxygen treatment of neuropsychiatric disorders following traumatic brain injury. Chin Med J (Engl), 2006. 119(23): p. 1978-82.
- Wright, J.K., et al., Case report: Treatment of mild traumatic brain injury with hyperbaric oxygen. Undersea Hyperb Med, 2009. 36(6): p. 391-9.
- Grillo, S.L., et al., Non-invasive infra-red therapy (1072 nm) reduces beta-amyloid protein levels in the brain of an Alzheimer’s disease mouse model, TASTPM. J Photochem Photobiol B, 2013. 123: p. 13-22.
- Gkotsi, D., et al., Recharging mitochondrial batteries in old eyes. Near infra-red increases ATP. Exp Eye Res, 2014. 122: p. 50-3.
- Quirk, B.J., et al., Near-Infrared Photobiomodulation in an Animal Model of Traumatic Brain Injury: Improvements at the Behavioral and Biochemical Levels. Photomedicine and Laser Surgery, 2012. 30(9): p. 7.
- Naeser, M.A., et al., Significant Improvements in Cognitive Performance Post-Transcranial, Red/Near-Infrared Light-Emitting Diode Treatments in Chronic, Mild Traumatic Brain Injury: Open-Protocol Study. JOURNAL OF NEUROTRAUMA, 2014. 31: p. 10.
- Pu, H., et al., Omega-3 polyunsaturated fatty acid supplementation improves neurologic recovery and attenuates white matter injury after experimental traumatic brain injury. J Cereb Blood Flow Metab, 2013. 33(9): p. 1474-84.
- Lewis, M., P. Ghassemi, and J. Hibbeln, Therapeutic use of omega-3 fatty acids in severe head trauma. Am J Emerg Med, 2013. 31(1): p. 273 e5-8.
- Hasadsri, L., et al., Omega-3 fatty acids as a putative treatment for traumatic brain injury. J Neurotrauma, 2013. 30(11): p. 897-906.
Disclaimer: I am not a medical doctor. I am not giving medical advice, diagnosis or treatment recommendations. The posts on this blog are my opinion. If you are thinking of following or using any of this information for any health related conditions, I would recommend you talk to your physician and seek guidance and help. I try to be as meticulous as possible in the information I use for these posts. I look for potential therapies that are low-risk/high impact. There are no guarantees, but knowledge is power and self-direction can lead you to uncover and do incredible things.
Source: Brain Injury and Sex: What Happens After a TBI? | Brain Health & Healing Foundation




 What is the largest sex organ in the body?
What is the largest sex organ in the body? The function of the pituitary is diverse, as it can affect a number of really important functions:
The function of the pituitary is diverse, as it can affect a number of really important functions:
