A legacy resource from NICHCY
Disability Fact Sheet 18 (FS18)
Links updated, August 2014
In This Publication:
Posted by Kostas Pantremenos in Tele/Home Rehabilitation, Video Games/Exergames on June 30, 2015
Abstract
This paper presents initial concepts from the REHAB@HOME project investigating the patient-centered design of game environments aimed to raising patients’ motivation and compliance with motor-cognitive rehabilitation programs. During the initial phase of the project a patient’s client was developed to deploy five rehabilitation games through main gaming platforms and interaction devices (Kinect, LeapMotion, Sifteo Cubes). Also, a professional client was designed to enable clinicians the remote monitoring of patients’ progress in home settings. We discuss main features developed for both clients that can inform the future realization of game-based solutions for upper body rehabilitation programs.
via A Game-Based Solution for In-Home Rehabilitation – Springer.
Posted by Kostas Pantremenos in Tele/Home Rehabilitation on June 30, 2015
Abstract
Stroke is often a severe and debilitating event that requires ongoing rehabilitation. The Community Stroke Rehabilitation Teams (CSRT) offer home-based stroke rehabilitation to individuals for whom further therapy is unavailable or inaccessible.
The objective of this study was to evaluate the cost-effectiveness of the CSRT program compared with a ‘No Therapy’ cohort. Data were collected on CSRT clients from January 2012 to February 2013. Comparator data were derived from a study of stroke survivors with limited access to rehabilitation.
Literature derived values were used to inform a long-term projection. Using Markov modelling, we projected the model for 35 years. One-way, two-way, and Probabilistic Sensitivity Analyses were performed. Results demonstrate that the CSRT has a Net Monetary Benefit of $43,115 over No Therapy, and is both less costly and more effective. The CSRT model of care should be considered when evaluating potential stroke rehabilitation delivery methods.
Posted by Kostas Pantremenos in Paretic Hand, Tele/Home Rehabilitation on June 30, 2015
 Abstract
Abstract
This single-case, mixed-method study explored the feasibility of self-administered, home-based SMART (sensorimotor active rehabilitation training) Arm training for a 57-yr-old man with severe upper-limb disability after a right frontoparietal hemorrhagic stroke 9 mo earlier. Over 4 wk of self-administered, home-based SMART Arm training, the participant completed 2,100 repetitions unassisted. His wife provided support for equipment set-up and training progressions.
Clinically meaningful improvements in arm impairment (strength), activity (arm and hand tasks), and participation (use of arm in everyday tasks) occurred after training (at 4 wk) and at follow-up (at 16 wk). Areas for refinement of SMART Arm training derived from thematic analysis of the participant’s and researchers’ journals focused on enabling independence, ensuring home and user friendliness, maintaining the motivation to persevere, progressing toward everyday tasks, and integrating practice into daily routine.
These findings suggest that further investigation of self-administered, home-based SMART Arm training is warranted for people with stroke who have severe upper-limb disability.
Posted by Kostas Pantremenos in Gait Rehabilitation - Foot Drop on June 30, 2015
Abstract
Introduction: Stroke is the leading cause of disability in adults. The correlations of balance and gait parameters are important for assessing and rehabilitating these patients. The purpose of this study was to compare the sense of balance by center of pressure (COP) sway and its relationship with gait parameters and functional independence in left (LH) and right (RH) chronic stroke patients. Twenty-one hemiparetic stroke patients treated at the hemiplegia outpatient clinic were invited to participate in this cross-sectional study. Patients were assessed for dominance, spasticity, injury time, muscle strength, and Functional Independence Measure (FIM). The COP sway was assessed with a force platform. For gait evaluation, patients were asked to walk along a preestablished path in the Motion Analysis Laboratory. The amplitudes of the COP in the anteroposterior (RH- 3.0±1.4 cm and LH-3.1±1.2) and mediolateral (RH- 1.7±1.2 and LH-1.5±0.5 cm) directions were similar in both groups. The parameters considered were length of steps and stride, single stance, gait velocity, and cadence.
Results: only the temporal parameters showed any statistically significant differences. All patients spent more time in the stance phase for the healthy lower limb (LH; p=0.0004; RH; p=0.001), specifically the single stance time. There was no difference in the performance of balance, gait, and functional independence among groups of chronic hemiparetic stroke patients when comparing left and right hemisphere lesions. The LH showed a significant (p<0.05) correlation between stride length, step length, and gait velocity with COP velocity sway for the healthy and paretic lower limbs. In both groups the area of COP was significantly correlated with stride length. Motor FIM was significantly correlated (r=-0.59, p<0.05) with the COP area sway in the left hemiparesis group.
Conclusion: There was no difference in the performance of balance, gait, and functional independence between groups. The left hemiparesis group showed a better correlation of COP and gait parameters than did the right hemiparesis group. The correlation of the COP sway area with stride length in both groups can serve as a guideline in the rehabilitation of these patients where training the static balance may reflect the improvement of the stride length.
Posted by Kostas Pantremenos in Constraint induced movement therapy CIMT, Recovery Plateau on June 30, 2015
Abstract
An open question in stroke rehabilitation is, if and how chronic patients can still make improvements after they reached a plateau in motor recovery. Previous research has shown that Constraint-Induced Movement Therapy (CIMT) might be effective in treating hemiparesis and supporting functional improvements in chronic patients, but that it might also be associated with higher costs in terms of demand, resources and inconvenience for the patient.
Here, we offer a new therapeutic approach that combines CIMT with a positive reinforcement component. We suggest that this new therapy, called Reinforcement Induced Movement Therapy (RIMT), might be similarly effective as CIMT and could be suitable for a broader population of chronic stroke patients.
We first implemented a computational model to study the potential outcome of different CIMT and RIMT therapy combinations. Then we present the results of an ongoing clinical trial that supports predictions from the model. We conclude that an optimally combined CIMT and RIMT therapy might propose a novel and powerful rehabilitation approach, addressing the specific needs of chronic stroke patients.
Posted by Kostas Pantremenos in Caregivers, Epilepsy on June 30, 2015
 When someone is having is seizure, they will often experience involuntary movement, changes in behavior, and awareness for lasting a couple minutes to an hour. If you’ve never witnessed a seizure, you might be shocked, confused, scared, worried, or all of the above. The most important thing, however, is for you to remain calm.
When someone is having is seizure, they will often experience involuntary movement, changes in behavior, and awareness for lasting a couple minutes to an hour. If you’ve never witnessed a seizure, you might be shocked, confused, scared, worried, or all of the above. The most important thing, however, is for you to remain calm.
Know the circumstances under which you should call for emergency medical services. If you are concerned for the person’s safety and feel unable to help the person, you should call for emergency medical attention, but in many cases, a person having a seizure will not need emergency assistance. Often, a person will have medication with them. Ask when you can. This is helpful and can be useful to them right away. Get emergency assistance if:
Continue —> How to Help Someone Who Is Having a Seizure | Cancer Facts Central.
Posted by Kostas Pantremenos in Neuroplasticity on June 30, 2015
JUNE 29, 2015 ~ BRAIN INJURY BLOG TORONTO
BY: SOPHIA VOUMAKIS
When I suffered my traumatic brain injury (TBI) in 2011, I believed that the cells in my brain which had been damaged were irreparable. But recent research suggests that the brain can repair itself, and that what was once damaged may be able to heal.
Before we explore the scientific evidence, we need to have a rudimentary understanding of how the brain works. Very simply, the brain is made up of two main groups of cells, neurons and glial. When these cells work together, the electrical activity they create enables us to move, think, emote, feel, remember – essentially, to live.
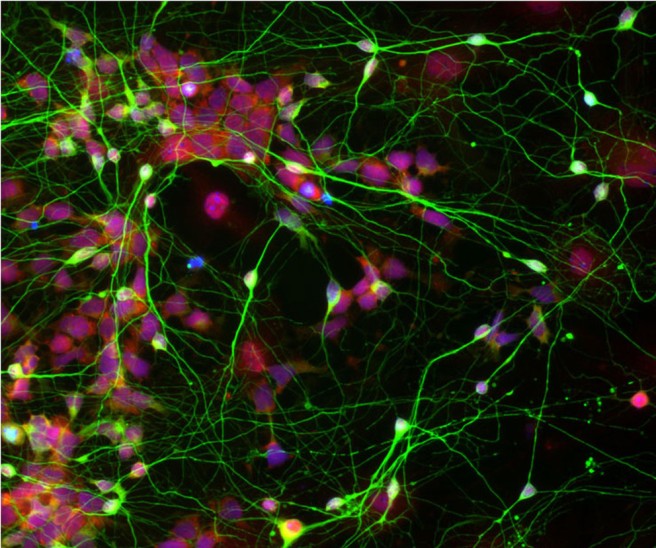 Human neural stem cells (red), originally reprogrammed from adult skin cells, functioning efficiently with brain cells (green). Photo credit: Chen Lab, Penn State University via Pen State News
Human neural stem cells (red), originally reprogrammed from adult skin cells, functioning efficiently with brain cells (green). Photo credit: Chen Lab, Penn State University via Pen State News
However when one of these cells gets damaged or dies, the result is damaged wiring and connections. If the damaged nerve is a motor neuron, then motor functioning is impaired. If the myelin cell is damaged, diseases such as Multiple Sclerosis (MS) develop.
Speaking at a TED Conference in July 2013, Dr. Siddharthan Chandran, director of the Centre for Clinical Brain Sciences, describes a case of a patient with MS whose brain scan showed myelin damage. Subsequent scans showed some repair in the area of the brain which had originally displayed damage. This repair had occurred with no medical intervention, which led Dr. Chandran to conclude that “the brain can repair itself, just not well enough.”
Dr. Chandran believes this discovery will lead to a new direction in finding therapies to treat brain disorders. Human stem cells, which can can self-renew to create new bone or liver cells, show great promise in this endeavour. Scientists hope that one day stem cells can be used to create new motor nerve or myelin cells.
In 2006, Japanese scientist Dr. Shinya Yamanaka discovered that just four ingredients can convert any adult cell into a master stem cell. This means that scientists can create a stem cell from any of us, and then make that cell relevant to our disease or injury, such as a motor neuron or a myelin cell. Yamanaka won a Nobel Prize for his work in 2012.
A recent clinical trial by Dr. Chandran, in collaboration with other scientists, illustrates this point. Researchers took stem cells from the bone marrow of patients with MS, grew myelin cells in the lab, and then injected them back into the patients’ veins. To measure whether the intervention was successful, the scientists examined the optic nerve. The size of the optic nerve was measured before the injection of the lab grown myelin cells, three and six months post injection (patients with MS usually have vision problems). Results showed the optic nerve had stopped shrinking, which Dr. Chandran believes is the result of the injected myelin cells, which promoted the brain’s own stem cells to do their job of laying down more myelin.
In addition, scientists can now use human cells to find ways to promote and activate the stem cells, which are already in our brain, to repair damage. Dr. Chandran believes this technique could replace animal testing in the future.
Although Dr. Chandran discussed these new advances in the treatment of brain disorders such as MS and Parkinson’s, these scientific advances may have applicability to the brain injury as well. To quote Dr. Chandran, “the day we can repair the damaged brain is sooner than we think.”
Since her TBI in 2011, Sophia has educated herself about TBI. She is interested in making research into TBI accessible to other survivors.
via Can the brain repair itself? | Brain Injury Blog TORONTO.
Posted by Kostas Pantremenos in Caregivers on June 29, 2015

A legacy resource from NICHCY
Disability Fact Sheet 18 (FS18)
Links updated, August 2014
In This Publication:
Continue —> Traumatic Brain Injury | Center for Parent Information and Resources.
Posted by Kostas Pantremenos in Paretic Hand on June 27, 2015
Abstract
This paper proposes an EMG based learning approach for estimating the displacement along the 2-axes (abduction/adduction and flexion/extension) of the human wrist in real-time. The algorithm extracts features from the EMG electrodes on the upper and forearm and uses Support Vector Regression to estimate the intended displacement of the wrist. Using data recorded with the arm outstretched in various locations in space, we train the algorithm so as to allow robust prediction even when the subject moves his/her arm across several positions in space. The proposed approach was tested on five healthy subjects and showed that a R 2 index of 63.6% is obtained for generalization across different arm positions and wrist joint angles.
Posted by Kostas Pantremenos in Paretic Hand, Spasticity on June 27, 2015
Abstract
Introduction/Background: In a Phase III, randomized, double-blind placebo-controlled study conducted in 34 sites from 9 countries, two doses of abobotulinumtoxinA (Dysport®) 500 and 1000 units (U) were shown to be efficacious on muscle tone for the treatment of hemiparetic adults post stroke or traumatic brain injury (TBI) with a favourable safety profile.1.
Materials and Methods: 243 patients received abobotulinumtoxinA 500 or 1000 U or placebo by intramuscular injection into their primary targeted muscle group (PTMG, selected from extrinsic finger flexors, wrist flexors and elbow flexors) and at least two other upper limb muscles, including shoulder muscles. Treatment was administered in a volume of 5.0 mL using electrostimulation. Doses administered to upper limb muscles are reported here.
Results: For the abobotulinumtoxinA 500 U group, mean (SD) doses (U) administered in fingers flexors were: 93.5 (17.0) for flexor digitorium profundus (FDP), 95.4 (14.3) for flexor digitorium superficialis (FDS) and 76.9 (26.8) for other finger flexors (flexor pollis longus, adductor pollicis); in wrist flexors: 92.2 (18.1) for flexor carpi radialis (FCR) and 89.9 (25.7) for flexor carpi ulnaris (FCU); in elbow flexors: 88.3 (28.5) for brachioradialis, 148.5 (60.2) for brachialis and 108.6 (49.5) for other elbow muscles (biceps brachii, pronator teres) and 122.2 (44.1) in shoulder muscles (triceps brachii, pectoralis major, subscapularis, latissimus dorsi). For the abobotulinumtoxinA 1000 U group, doses administered were 195.5 (25.9) for FDP, 196.8 (28.4) for FDS, 157.0 (53.3) for other finger flexors, 178.1 (45.5) for FCR, 171.2 (45.2) for FCU, 172.1 (44.8) for brachioradialis, 321.4 (103.2) for brachialis, 216.5 (92.2) for other elbow muscles and 300.0 (129.1) in shoulder muscles.
Conclusion: In this Phase III worldwide study in hemiparetic patients with upper limb spasticity post stroke/TBI, mean doses administered were 76.9–196.8 U for muscles in the finger flexors, 89.9-178.1 U for muscles in wrist flexors, 88.3–321.4 U for muscles in the elbow flexors and 122.2–300.0 U in shoulder muscles. Total dose administered (in the PTMG and at least 2 upper limb muscles) was 500 or 1000 U, which was previously shown to improve muscle tone in this patient population.