Posts Tagged Wrist
[Abstract] A Robotic Hand for Rehabilitation of Wrist and Fingers
Posted by Kostas Pantremenos in Paretic Hand, Rehabilitation robotics on January 20, 2024
Abstract
Carpal Tunnel Syndrome is a common neural disease among people with repetitive wrist movements. It affects the sensation and movement of the thumb, index finger, middle finger, and half of the ring finger. The syndrome can be treated effectively through therapeutic exercises in the early stages of diagnosis or through surgery in more severe cases. However, the lack of physiotherapists poses a significant challenge in hospitals where thorough observation is required. The proposed rehabilitation robot focuses on wrist and finger exercises designed explicitly for Carpal Tunnel Syndrome and intended for clinical use. The sensors detect the patient’s range of motion and display the data for the therapist, who can then determine the appropriate exercises. Finger exercises include flexion-extension, mimicking grasping patterns, while wrist exercises include flexion-extension and hyperextension. The rehabilitation program consists of three stages and several iterations, progressing according to the patient’s recovery, which can be observed through the data collected from the sensors. The rehabilitation robot utilizes an under-actuation method that drives proximal interphalangeal, and distal interphalangeal joints of the four fingers using two servo motors for finger rehabilitation. The thumb is actuated using a separate servo motor. A stepper motor actuates wrist movement. A mathematical model was developed to simulate the movements of the linkage system, which is deployed in the sagittal plane of the fingers. The effectiveness of the rehabilitation robot has been verified through simulations and experiments.
[Abstract] Preliminary Results from a Six-Week Home-Based Evaluation of a Rehabilitation Device for Hand and Wrist Therapy After Stroke
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION, Rehabilitation robotics, Tele/Home Rehabilitation on November 17, 2023
Abstract
While many robot-aided solutions have been proposed for the rehabilitation of the distal segment of the upper limb, very few take into account the synergy between the wrist and fingers to allow them to train simultaneously in a home environment. WiGlove is a passive robotic orthosis designed to address this need. This wearable, wireless device enables stroke survivors to perform flexion/extension exercises of both the wrist and fingers while performing ADL or playing therapeutic computer games. As a part of its user-centred design process, this paper presents a case study of a 6-week feasibility evaluation of the WiGlove conducted at a stroke survivor’s home without assistance from the therapists. The participant trained with the device for an average of 48 minutes a day and showed a noticeable reduction in the spasticity of the fingers and improved performance in the box and block test. He expressed satisfaction with its usability and suitability for the home environment. These results show overwhelmingly positive outcomes in terms of its acceptance, usability and effectiveness in offering home-based rehabilitation of the wrist and fingers.
[REVIEW] A Review of Parallel Robots: Rehabilitation, Assistance, and Humanoid Applications for Neck, Shoulder, Wrist, Hip, and Ankle Joints
Posted by Kostas Pantremenos in REHABILITATION, Rehabilitation robotics on November 7, 2023
Abstract
This review article presents an in-depth examination of research and development in the fields of rehabilitation, assistive technologies, and humanoid robots. It focuses on parallel robots designed for human body joints with three degrees of freedom, specifically the neck, shoulder, wrist, hip, and ankle. A systematic search was conducted across multiple databases, including Scopus, Web of Science, PubMed, IEEE Xplore, ScienceDirect, the Directory of Open Access Journals, and the ASME Journal. This systematic review offers an updated overview of advancements in the field from 2012 to 2023. After applying exclusion criteria, 93 papers were selected for in-depth review. This cohort included 13 articles focusing on the neck joint, 19 on the shoulder joint, 22 on the wrist joint, 9 on the hip joint, and 30 on the ankle joint. The article discusses the timeline and advancements of parallel robots, covering technology readiness levels (TRLs), design, the number of degrees of freedom, kinematics structure, workspace assessment, functional capabilities, performance evaluation methods, and material selection for the development of parallel robotics. It also examines critical technological challenges and future prospects in rehabilitation, assistance, and humanoid robots.
Keywords:
assistance; exoskeletons; parallel robots; prosthetics; rehabilitation
1. Introduction
Rehabilitation and assistance for human body joints play crucial roles in people’s health, well-being, and quality of life. These activities are essential for helping individuals recover or improve their functionality, mobility, and autonomy following injuries, surgeries, or diseases affecting the joints. In recent years, technological advances have opened up new possibilities in the use of parallel robots in the fields of rehabilitation, assistive technologies, and humanoid systems. These robotic systems are specifically designed to facilitate the recovery of motor and functional skills. Featuring a parallel mechanical structure, these robots offer greater precision, stability, and adaptability to meet the individual rehabilitation or assistance needs of patients.
Rehabilitation technologies aid in the recovery or improvement of motor function after an injury or illness. Utilizing parallel robots in rehabilitation enables specialists to help patients regain strength and mobility in affected joints more rapidly and efficiently than traditional therapeutic methods. The scope of these technologies includes rehabilitation devices for patients with head and neck injuries [1,2,3,4]; pediatric rehabilitation devices for the arm [5]; wearable rehabilitation devices for the arm [6]; exoskeletons for the arms of patients with stroke and spinal cord injuries [7]; and devices specifically designed for wrist [8,9,10], ankle [11,12,13,14,15,16], and foot rehabilitation [17].
Parallel robots in assistive devices help individuals carry out activities of daily life, thereby providing greater autonomy and independence. Examples include prostheses designed to offer functional mobility by replacing missing limbs in amputees [18], as well as shoulder disarticulation arm prostheses [19,20], wrist prostheses [21], and disarticulated hip prostheses [22].
The integration of humanoid robots into medical rehabilitation and assistance offers exciting opportunities. These include personalized therapy, precise motivation and tracking, assisted mobility, and the objective assessment of patients’ progress.
Parallel robots have advanced significantly in the fields of architectural [23] and mathematical modeling, particularly in kinematic [24,25,26] and dynamic analyses [27]. These robots can move at high speeds due to their lightweight and simple structural design [28]. They are also highly rigid, making them ideal for tasks requiring substantial force or pressure [29]. Their high-precision design minimizes unwanted movements and vibrations [30]. Furthermore, they can support heavier loads [31,32] and offer better positioning accuracy due to their high rigidity and low weight, which ensure minimal deformation [33]. Utilizing sensor technology and control algorithms, these parallel robots can automatically adjust to meet each patient’s specific needs, whether for rehabilitation or assistance, thus delivering a highly personalized experience.
However, there is a gap in the current literature concerning the use of parallel robots in the fields of rehabilitation, assistance, and humanoid systems. This review aims to address this gap by summarizing the available evidence on the utilization of parallel robots.
Therefore, this article aims to review the state of parallel robot technology as applied to rehabilitation, assistance, and humanoid systems, focusing on joints with three degrees of freedom in the human body: the neck, shoulder, wrist, hip, and ankle. Initially, the search strategy, inclusion and exclusion criteria, quality assessment, and data extraction methods are defined. Subsequently, the search results are presented, along with a detailed description of the biomechanics of the joints, parallel robots, and applications in the medical field. The article then discusses the timeline and advancements of parallel robots between 2012 and 2023 and examines the technology readiness levels (TRLs), design, number of degrees of freedom, kinematics structure, workspace assessment, functional capabilities, performance methods, and material selection in the development of parallel robotics, as well as the critical technological challenges and future prospects in rehabilitation, assistance, and humanoids. Finally, conclusions are presented. […]
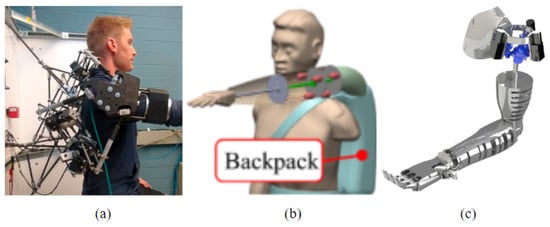
[Abstract + References] A State of Art Overwiew on Wrist Rehabilitation Exoskeletons – Conference paper
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION, Rehabilitation robotics on April 21, 2023
Abstract
Exoskeleton-type robotic systems have been of great interest in scientific research in recent years, due to the many applications in which they can be used. They can be found in industry to enhance the physical abilities of the human operator such as speed and strength, in the military, where in addition to enhancing physical abilities, it also provides protection for the operator and in the medical field, in rehabilitation applications where it provides support. and assistance in performing various movements. In this paper, the focus is on exoskeleton robotic devices used to rehabilitate the upper limb for patients recovering from a stroke. There will be a review in the literature for the latest devices developed, they will be classified according to the number of degrees of freedom rendered and respectively the type of drive system used.
References
- Puthenpurakal, A., Crussell, J.: Stroke 1: definition, burden, risk factors and diagnosis. In: Nursing Times, (2017)Google Scholar
- Go, A.S., Mozaffarian, D., Roger, V.L., Benjamin, E.J., Berry, J.D., Borden, W.B., et al.: Heart disease and stroke statistics. In: American heart association, (2016)Google Scholar
- Townsend, K.W.N., Bhatnagar, P., Smolina, K., Nichols, M., Leal, J., Luengo-Fernandez, R. Rayner, M.: Coronary heart disease statistics. In: British heart foundation, (2012)Google Scholar
- Rehmat, N., Zuo, J., Meng, W. et al.: Upper limb rehabilitation using robotic exoskeleton systems: a systematic review. Int. J. Intell. Robot. Appl., (2018)Google Scholar
- Chen, J., Zhu, Q., Yu, L., Li, Y., Jia, S., Zhang, Z.: Stroke risk factors of stroke patients in China: A nationwide community-based cross-sectional study. Int. J. Environ. Res. Public Health, (2022)Google Scholar
- Townsend, N., Nichols, M., Luengo-Fernandez, R., Leal, J., Gray, A., Scarborough, P., Rayner, M.: European cardiovascular disease statistics. Eur. Hear. Netw. AISBL, (2012)Google Scholar
- Tsugawa, Y., Kumamaru, H., Yasunaga, H., Hashimoto, H., Horiguchi, H., Ayanian, J. Z.: The association of hospital volume with mortality and costs of care for stroke in Japan. In: National Library of Medicine, (2013)Google Scholar
- http://www.stroke.org.nz/stroke-facts-and-fallacies—Accessed in 20 June 2022.
- Lindsay, M.P., Norrving, B., Sacco, R.L., Brainin, M., Hacke, W., Martins, S., Pandian, J., Feigin, V.: Global stroke fact sheet. In: National library of Medicine, (2019)Google Scholar
- https://www.safestroke.eu/wp-content/uploads/2020/10/01-At_What_Cost_EIOS_Factsheet.pdf Accesed in 01 July 2022
- https://www.socialstyrelsen.se/globalassets/sharepointdokument/artikelkatalog/statistik/2021-12-7644.pdf. Accesed in 01 July 2022
- Gull, M.A., Bai, S., Bak, T.: A review on design of upper limb exoskeletons. Robotics, (2020)Google Scholar
- Gopura, R.C., Kiguchi, K., Bandara, S.V.: A brief review on upper extremity robotic exoskeleton systems. In: Industrial and information systems, (2011)Google Scholar
- Pezent, E., Rose, C., Deshpande, A., Omalley, M.: Design and characterization of the OpenWrist: A robotic wrist exoskeleton for coordinated hand-wrist rehabilitation. In: International conference on rehabilitations robotics IEEE Xplore, (2017)Google Scholar
- Esmaeili, M., Dailey, W., Burdet, E., Campolo, D.: Ergonomic design of a wrist exoskeleton and its effects on natural motor strategies during redundant tasks. In: IEEE International conference on robotics and automation, (2013)Google Scholar
- Kazerooni, H.: Exoskeletons for human performance augmentation. In: Springer hand- book of robotics, (2008)Google Scholar
- Song, Z., Guo, S.: Design process of exoskeleton rehabilitation device and implemen tation of bilateral upper limb motor movement. J. Med. Biol. Eng., 2011Google Scholar
- Meng, W., Sheng, B., Klinger, M., Liu, Q., Zhou, Z., Xie, S.Q.: Design and control of a robotic wrist orthosis for joint rehabilitation. In: IEEE/ASME (AIM) International conference on advanced intelligent mechatronics, (2015)Google Scholar
- Ates, S., Mora-Moreno, I., Wessels, M., Stienen, A.: Combined active wrist and hand orthosis for home use: Lessons learned. In: Conference: Rehabilitation robotics (ICORR), (2015)Google Scholar
- Ates, S., Lobo-Prat, J., Lammertse, P.,vad der Koji, H., Stenen, A.H.A.: SCRIPT passive orthosis: Design and technical evaluation of the wrist and hand orthosis for rehabilitation training at home. In: IEEE international conference on rehabilitation robotics, (2013)Google Scholar
- Ates, S., Haarman, C.J., Stenen, A.H.A.: SCRIPT passive orthosis: design of interactive hand and wrist exoskeleton for rehabilitation at home after stroke. Auton. Robot., (2017)Google Scholar
- Nam, H.S., Koh, S., Kim, Y.J., Beom, J., Lee, W.H., Lee, S.U., Kim, S.: Biomechanical reactions of exoskeleton neurorehabilitation robots in spastic elbows and wrists. In: IEEE Transactions on neural systems and rehabilitation engineering, (2017)Google Scholar
- Hussain, S., Jamwal, P.K., Van Vliet, P., Ghyesh, M.H.: State of the art robotic devices for wrist rehabilitation: Design and control aspects. In: IEEE Transactions on human-machine systems, (2020)Google Scholar
- Lambelet, C., Lyu, M., Wenderoth, N., Woolley, D., Gassert, R.: The eWrist—A wearable wrist exoskeleton with sEMG-based force control for stroke rehabilitation. In: IEEE International conference on rehabilitation Robotics, (2017)Google Scholar
- Visconti, P., Gaetani, F., Zappatore, G.A., Primiceri, P.: Technical features and functionalities of Myo armband: An overview on related literature and advanced appli cations of myoelectric armbands mainly focused on arm prostheses. Int. J. Smart Sens. Intell. Syst., 2018Google Scholar
- Lambelet, C., Temiraliuly, D., Siegenthaler, M., Wirth, M., Woolley, D.G., Lambercy, O., Gassert, R., Wenderoth, N.: Characterization and wearability evaluation of a fully porta ble wrist exoskeleton for unsupervised training after stroke. J. NeuroEngineering Rehabil., (2020)Google Scholar
- Xiao, Z.G., Menon, C.: Towards the development of a portable wrist exoskeleton. In: IEEE international conference on robotics and biomimetics, (2011)Google Scholar
- Andrikopoulos, G., Nikolakopoulos, G., Manesis, S.: Motion control of a novel robotic wrist exoskeleton via pneumatic muscle actuators. In: 20th IEEE international conference on emerging technologies and factory automation (ETFA), (2015)Google Scholar
- Al-Fahaam, H., Davis, S., Nefti-Meziani, S.: Wrist rehabilitation exoskeleton robot based on pneumatic soft actuators. In: Proceeding of international conference for stu dents on applied engineering (ICSAE), (2017)Google Scholar
- Higuma, T., Kiguchi, K., Arata, J.: Low-profile two-degree-of-freedom wrist exoskel eton device using multiple spring blades. IEEE Robot. Autom. Lett., (2017)Google Scholar
- Lee, J., Song, B.W., Yang, W.: Design of exoskeleton-type wrist human–machine inter- face based on over-actuated coaxial spherical parallel mechanism. Adv. Mech. EngGoogle Scholar
- Del Cerro, D.S., Copaci, D., E Moreno, L., Blanco, D.: SMA based wrist exoskeleton for rehabilitation therapy*. IROS, (2018)Google Scholar
- Dragusanu, M., Lisini, T., Iqbal, M.Z., Prattichizzo, D., Melvezzi, M.: Design, development, and control of a tendon-actuated exoskeleton for wrist rehabilitation and Training. In: IEEE international conference on robotics and automation (ICRA), (2020)Google Scholar
- Gupta, A., O’Malley, M.K., Patoglu, V., Burgar, C.: Design, control and performance of rice wrist: A force feedback wrist exoskeleton for rehabilitation and training. Int. J. Robot. Res., (2008)Google Scholar
- Ren, Y., Kang, S.H., Park, H.S., Wu, Y.N., Zhang, L.Q.: Developing a multi-joint upper limb exoskeleton robot for diagnosis, therapy, and outcome evaluation in Neuro rehabilitation. In: IEEE transactions of neural systems and rehabilitation engineer ing, (2013)Google Scholar
- Troncossi, M., Mozaffari Foumashi, M., Mazzotti, C., Zannoli, D., Castelli, V.P.: Design and manufacturing of a hand-and-wrist exoskeleton prototype for the rehabilitation of post-stroke patients. In: Quaderni del DIEM—GMA. Atti della Sesta Giornata di Studio Ettore Funaioli, (2012)Google Scholar
- Rahman, M.H., Rahman, M.J., Cristobal,O.L., Saad, M., Kenne, J.P, Archambault, P.S.: Development of a whole arm wearable robotic exoskeleton for rehabilitation and to assist upper limb movements. Robotica, (2014)Google Scholar
- Bartlett, N.W., Lyau, V., Raiford, W.A., Holland, D., Gafford, J.B., Ellis, T.D., Walsh, C.J.: A soft robotic orthosis for wrist rehabilitation. In: J. Med. Devices, ( 2015)Google Scholar
- Buongiorno, D., Sotgiu, E., Leonardis, D., Marchschi, S., Solassi, M., Frisoli, A.: WRES: A novel 3 DoF wrist exoskeleton with tendon-driven differential transmission for neuro-rehabilitation and teleoperation. In: IEEE robotics and automation letters, (2018)Google Scholar
- Yang, S., Li, M., Wang, J., Wang, T., Liang, Z., He, B., Xie, J., Xu, G.: A novel wrist reha- bilitation exoskeleton using 3D-printed multi-segment mechanism. In: 43rd Annual international conference of the IEEE engineering in medicine & biology society (EMBC), (2021)Google Scholar
- Shi, K., Song, A., Li, Y., Li H., Chen, D., Zhu, L.: A cable-driven three-DOF wrist rehabilitation exoskeleton with improved performance. Front. Neuro Robot., (2021)Google Scholar
[WEB] Formative Usability Evaluation of WiGlove – A Home-based Rehabilitation Device for Hand and Wrist Therapy after Stroke
Posted by Kostas Pantremenos in Paretic Hand, Tele/Home Rehabilitation on March 14, 2023
ABSTRACT
WiGlove is a passive dynamic orthosis aimed at home-based post-stroke rehabilitation of the hand and wrist. This paper highlights results from WiGlove’s formative evaluation as the first step towards its deployment. In this study, twenty healthy participants evaluated the usability and safety of the WiGlove compared to its predecessor, the state-of-the-art SCRIPT Passive Orthosis (SPO). In this within-subject experiment, they performed various tasks such as donning/doffing, adjusting the tension, grasping, etc., with both gloves and rated them using a Likert scale-based questionnaire. The results showed improvements in several aspects of usability and safety. This study provides preliminary evidence of WiGlove’s fitness for the next assessment with its intended users, people recovering from stroke with sustained hand and wrist impairment.
Supplemental Material
HRI_paper_video_scaled_720p_HVEC.mp4
This video presents the results of the usability evaluation of the WiGlove. It is a passive dynamic orthosis designed to assist stroke survivors to perform home-based rehabilitation of their hand and wrist to regain their ability to perform activities of daily life (ADL). It allows hemiparetic stroke survivors to perform flexion exercises while playing interactive games on a tablet and while performing activities of daily life without the assistance of the therapist. This was developed based on the knowledge gained from its predecessor, SCRIPT Passive Orthosis (SPO) and to address its functional and usability limitations. Comparative evaluation by healthy participants found improvements in several aspects of WiGlove’s usability such as ease of donning/doffing, unrestricted ab/adduction of the wrist, suitability for ADL, safety and aesthetics compared to SPO.
References
- Hassanin Al-Fahaam, Steve Davis, Samia Nefti-Meziani, and Theo Theodoridis. 2018. Novel soft bending actuator-based power augmentation hand exoskeleton controlled by human intention. Intelligent Service Robotics 11, 3 (2018), 247–268.
- Farshid Amirabdollahian, Serdar Ates, Angelo Basteris, Alfredo Cesario, Jaap Buurke, H Hermens, Dennis Hofs, Ellinor Johansson, Gail Mountain, Nasrin Nasr, et al. 2014. Design, development and deployment of a hand/wrist exoskeleton for home-based rehabilitation after stroke-SCRIPT project. Robotica 32, 8 (2014), 1331–1346.
- Andrew A Amis. 1987. Variation of finger forces in maximal isometric grasp tests on a range of cylinder diameters. Journal of biomedical engineering 9, 4 (1987), 313–320.
- Serdar Ates, Beatriz Leon, Angelo Basteris, Sharon Nijenhuis, Nasrin Nasr, Patrizio Sale, Alfredo Cesario, Farshid Amirabdollahian, and Arno HA Stienen. 2014. Technical evaluation of and clinical experiences with the SCRIPT passive wrist and hand orthosis. In 2014 7th International Conference on Human System Interactions (HSI). IEEE, 188–193.
- Joni G Barry, Sandy A Ross, and Judy Woehrle. 2012. Therapy incorporating a dynamic wrist-hand orthosis versus manual assistance in chronic stroke: A pilot study. Journal of Neurologic Physical Therapy 36, 1 (2012), 17–24.
- Alberto Borboni, Maurizio Mor, and Rodolfo Faglia. 2016. Gloreha-hand robotic rehabilitation: Design, mechanical model, and experiments. Journal of Dynamic Systems, Measurement, and Control 138, 11 (2016).
- Tobias Bützer, Olivier Lambercy, Jumpei Arata, and Roger Gassert. 2021. Fully wearable actuated soft exoskeleton for grasping assistance in everyday activities. Soft robotics 8, 2 (2021), 128–143.
- Tianyao Chen and Peter S Lum. 2016. Hand rehabilitation after stroke using a wearable, high DOF, spring powered exoskeleton. In 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). IEEE, 578–581.
- Hermano Igo Krebs, Bruce T Volpe, Dustin Williams, James Celestino, Steven K Charles, Daniel Lynch, and Neville Hogan. 2007. Robot-aided neurorehabilitation: a robot for wrist rehabilitation. IEEE transactions on neural systems and rehabilitation engineering 15, 3 (2007), 327–335.
- Gert Kwakkel. 2009. Intensity of practice after stroke: More is better. Schweizer Archiv fur Neurologie und Psychiatrie 160, 7 (2009), 295–298.
- Beatriz Leon, Angelo Basteris, Francesco Infarinato, Patrizio Sale, Sharon Nijenhuis, Gerdienke Prange, and Farshid Amirabdollahian. 2014. Grasps recognition and evaluation of stroke patients for supporting rehabilitation therapy. BioMed Research International 2014 (2014).
- Pawel Maciejasz, Jörg Eschweiler, Kurt Gerlach-Hahn, Arne Jansen-Troy, and Steffen Leonhardt. 2014. A survey on robotic devices for upper limb rehabilitation. Journal of neuroengineering and rehabilitation 11, 1 (2014), 3.
- Bernardo Noronha and Dino Accoto. 2021. Exoskeletal devices for hand assistance and rehabilitation: A comprehensive analysis of state-of-the-art technologies. IEEE Transactions on Medical Robotics and Bionics 3, 2 (2021), 525–538.
- S Radhakrishnan and M Nagaravindra. 1993. Analysis of hand forces in health and disease during maximum isometric grasping of cylinders. Medical and Biological Engineering and Computing 31, 4 (1993), 372–376.
- Vignesh Velmurugan, Luke Wood, and Farshid Amirabdollahian. 2021. Requirements for a home-based rehabilitation device for hand and wrist therapy after stroke. In UKRAS21: The 4th UK Robotics and Autonomous Systems Conference. 23. https://doi.org/10.31256/Xw5Aj7Q
[ARTICLE] Laboratory-Based Examination of the Reliability and Validity of Kinematic Measures of Wrist and Finger Function Collected by a Telerehabilitation System in Persons with Chronic Stroke – Full Text
Posted by Kostas Pantremenos in Paretic Hand, Tele/Home Rehabilitation on March 5, 2023
Abstract
We have developed the New Jersey Institute of Technology—Home Virtual Rehabilitation System (NJIT—HoVRS) to facilitate intensive, hand-focused rehabilitation in the home. We developed testing simulations with the goal of providing richer information for clinicians performing remote assessments. This paper presents the results of reliability testing examining differences between in-person and remote testing as well as discriminatory and convergent validity testing of a battery of six kinematic measures collected with NJIT—HoVRS. Two different groups of persons with upper extremity impairments due to chronic stroke participated in two separate experiments. Data Collection: All data collection sessions included six kinematic tests collected with the Leap Motion Controller. Measurements collected include hand opening range, wrist extension range, pronation-supination range, hand opening accuracy, wrist extension accuracy, and pronation-supination accuracy. The system usability was evaluated by therapists performing the reliability study using the System Usability Scale. When comparing the in-laboratory collection and the first remote collection, the intra-class correlation coefficients (ICC) for three of the six measurements were above 0.900 and the other three were between 0.500 and 0.900. Two of the first remote collection/second remote collection ICCs were above 0.900, and the other four were between 0.600 and 0.900. The 95% confidence intervals for these ICC were broad, suggesting that these preliminary analyses need to be confirmed by studies with larger samples. The therapist’s SUS scores ranged from 70 to 90. The mean was 83.1 (SD = 6.4), which is consistent with industry adoption. There were statistically significant differences in the kinematic scores when comparing unimpaired and impaired UE for all six measures. Five of six impaired hand kinematic scores and five of six impaired/unimpaired hand difference scores demonstrated correlations between 0.400 and 0.700 with UEFMA scores. Reliability for all measures was acceptable for clinical practice. Discriminant and convergent validity testing suggest that scores on these tests may be meaningful and valid. Further testing in a remote setting is necessary to validate this process.
1. Introduction
Cerebrovascular accident (CVA) or stroke, is a leading cause of long-term disability in adults [1]. Despite more than a decade of investigating innovative approaches to rehabilitation, many individuals are left with upper limb deficits that hinder their ability to function independently [2]. Persistent, hand-related disability has a substantial effect on the productivity of the growing cohort of younger persons with milder strokes [3] and increases the cost of care for older and more impaired persons with strokes [4]. Current service delivery models emphasizing short, independence focused in-patient rehabilitation stays [5] and intermittent, low volume outpatient rehabilitation sessions [6] restrict the amount of rehabilitation a patient receives. This points to the need for rehabilitation services that patients can perform independently at home.
Recently, innovative telerehabilitation systems have been developed using information and communication technologies to provide rehabilitation services at a distance. A 2020 Cochrane Review of telerehabilitation interventions focusing on upper extremity function in persons with stroke demonstrated that persons receiving telerehabilitation made similar improvements in motor function as compared to in-person interventions [7]. We have developed the New Jersey Institute of Technology—Home Virtual Rehabilitation System (NJIT—HoVRS) to facilitate intensive, arm, wrist, hand, and finger-focused rehabilitation in the home [8]. The system integrates a Leap Motion Controller® (LMC) and a suite of custom designed simulations to provide rehabilitation of the upper extremity through engaging games. Preliminary studies of NJIT—HoVRS support that persons with stroke can use this system effectively in the home with minimal remote supervision [8] and that performing a 12-week program of hand and arm training results in meaningful improvements in hand and arm function [9,10].
A smaller body of literature exists examining the measurement of movement in a telerehabilitation setting. Small studies of telerehabilitation-based outcome measures suggest that shoulder and elbow motor function of persons with stroke can be measured meaningfully by performing standard clinical tests using camera-based approaches. These measurements are limited to performance-based tests and measurements of range of motion [11]. Similar to in-person testing, remotely collected measures that focus purely on range of motion or task outcomes fail to differentiate between recovery processes and the development of efficient compensatory strategies [12]. Several authors cite kinematic analysis as a means to identify the normalization of motor function in persons with stroke [12,13,14]. Therefore, reliable and valid kinematic measurements that can be collected at home may allow therapists to adjust plans of care in a manner that would emphasize the normalization of movement patterns, possibly making interventions prescribed based on these tests more effective [15]. To address this gap, we designed a set of kinematic measures of wrist and finger movement that can be conducted remotely in an attempt to add to the ability of clinicians to measure motor recovery as it occurred in persons using NJIT—HoVRS in a telerehabilitation environment [8].
Kinematic measures captured by NJIT—HoVRS are collected via the LMC, which utilizes a pair of cameras and a set of infrared light emitting diodes (LEDs). Images collected by the cameras are transmitted via USB to the LMC’s tracking software, which analyzes and transforms the images into three dimensional representations of these images. This allows for real time, camera-based estimation of wrist and finger angles and position measures without an expensive and cumbersome wearable apparatus. Published psychometrics of LMC based position and movement measurements have been mixed, with static measures generally performing better than dynamic measures [16,17,18]. Studies of the LMC with a more clinical focus demonstrate that joint specific wrist and finger position and range of motion data collected by the LMC correlated with measurements collected with an electro-goniometer in a population of healthy individuals in their 20s [19]. In another study, repetitive wrist and finger movements produced changes in position data that correlated well with impairment level in persons with Parkinson’s disease [20].
A study by Gieser et al. utilized finger positions and joint angles to classify hand gestures in children with cerebral palsy [21]. Taken together, these studies begin to suggest that the LMC produces measurements that may be sufficiently accurate for clinical purposes.
We developed a set of testing simulations to accompany NJIT—HoVRS’ intervention simulations to collect a battery of kinematic measures with the goal of providing richer information for clinicians performing remote assessments of their stroke patients. As a first step toward validating these kinematic measurements remotely at home, this paper will describe the results of two different studies examining: (1) reliability testing focused on the agreement between in laboratory and remotely collected measurements as well as a test of the system’s usability in the remote collection mode; and (2) discriminatory as well as convergent validity testing of a battery of six kinematic measures collected with the NJIT—HoVRS system in a laboratory setting. […]

[Abstract] Vagus Nerve Stimulation (VNS) Paired With Upper Extremity Rehabilitation In Chronic Stroke: Improvements In Wrist And Hand Impairment And Function
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION on February 11, 2023
Abstract
Introduction: In the VNS-REHAB trial, vagus nerve stimulation (VNS) paired with task-specific arm and hand rehabilitation (Paired VNS) led to clinically meaningful improvements in both impairment and function of the upper extremity in people with chronic ischemic stroke. In this post hoc analysis of trial data, we assessed whether improvements were driven by proximal (shoulder and elbow) and/or distal (wrist and hand) components of the Upper Extremity Fugl-Meyer Assessment (FMA-UE). We also wanted to determine whether FMA-UE improvements correlated with improvements in functional outcome.
Methods: Chronic stroke participants (n=108) with moderate-severe UE impairment (FMA-UE 35.1±8) were implanted with the VNS device and randomized to Paired VNS (n=53) or Controls (n=55). Participants underwent 6 weeks of in-clinic therapy followed by 90 days of a home exercise program. Outcomes, including the FMA-UE and Wolf Motor Function Test (WMFT), were collected after in-clinic therapy (Post-1) and 90 days of home therapy (Post-90).
Results: Distal FMA-UE change was significantly greater after Paired VNS compared to Controls, both at Post-1 (2.43±2.52 vs. 0.64±2.33; 95% CI 0.87-2.72, p<0.001, Cohen’s d=0.74) and Post-90 (2.58±2.88 vs. 0.93±3.16; 95% CI 0.5-2.81, p=0.005, Cohen’s d=0.55). Proximal FMA-UE change was also greater after Paired VNS compared to Controls but did not reach statistical significance at either timepoint. At Post-90, both proximal (r=0.50, p<0.0001) and distal (r=0.42, p=0.001) FMA-UE change significantly correlated with WMFT change after Paired VNS, but not in Controls (proximal: r=0.24, p=0.06; distal: r=0.14, p=0.24).
Conclusions: The data suggest that Paired VNS leads to greater improvement in the distal upper extremity compared to rehabilitation alone and that improvements in FMA-UE scores are correlated with improvements in function. These findings suggest that the improvements seen with Paired VNS are of functional importance. A greater understanding of region-specific arm and hand recovery after Paired VNS therapy will facilitate planning and implementation of a personalized neurorehabilitation approach based on patient-specific impairments.
[Abstract] Preliminary Design and Finite Element Analysis of A Low-Cost Wrist Rehabilitation Robot
Posted by Kostas Pantremenos in Paretic Hand, Rehabilitation robotics, Tele/Home Rehabilitation on February 6, 2023
Abstract:
Wrist rehabilitation robots are fundamental for helping patients with stroke or wrist injuries and also decrease the workload of physiotherapists. Though wrist rehabilitation robots are essential, recent wrist rehabilitation robots have shortcomings such as heavy weight, immobility, costliness, etc. To remedy these shortcomings, in this study, we developed and produced a low-cost and mobile robotic device for these patients with partial paralysis. The device is designed to assist the patient to perform wrist exercises comfortably in the home environment without being dependent on rehabilitation centers and/or physiotherapists. Therefore, we offer the device driven by a 3D-printed mechanism in two degrees of freedom. In addition, we analyzed and simulate the robot via finite element analysis and Solidworks respectively. The results indicate the robot provides enough force, torque, and range of motion. For these reasons, the robot can be used as a compact and lighter robot in hardware and a cheaper robot in cost. So it is feasible and affordable for real-time application in wrist rehabilitation.
[WEB] NHS trust levels up patient and therapist experience with new Hand Therapy app
Posted by Kostas Pantremenos in Apps, Paretic Hand on December 8, 2022
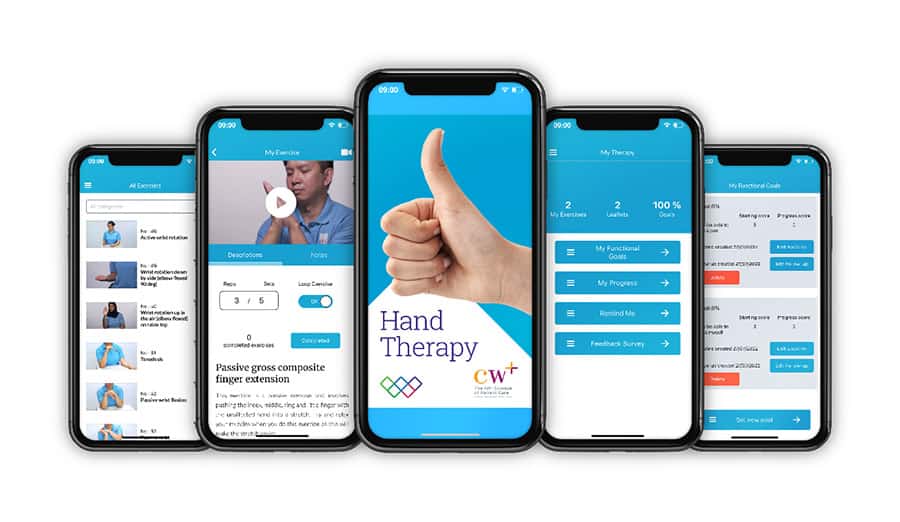
Chelsea and Westminster Hospital NHS Foundation Trust and its official charity, CW+, have launched a brand-new version of their smartphone app, Hand Therapy: The Exercise Prescription App, based on clinical and patient feedback.
The award-winning app provides patients with treatment information and a therapist-tailored exercise programme to aid recovery from hand and wrist injuries.
The improved version of the existing app has new functionality, content, and updated features to improve the patient and therapist experience.
Led by the trust’s senior occupational therapist (OT), Hayley Fay, and funded by CW+, the new version of the Hand Therapy app now offers 50 new videos. This has resulted in a library of over 130 instructional videos and audio tutorials, which patients can follow and replicate at home.
The updated app was developed based on patient and clinician feedback and will continue to be used in busy clinical settings. It remains accessible and free to download on a smartphone.
Hand Therapy updates will optimise existing features, enabling the hand therapy exercises to reorder in accordance with the prescription with additional ‘exercise repetitions’ and ‘completed’ buttons for each exercise. The catalogue has also been sub-categorised with an enhanced search function to make finding exercises quicker.
In addition, the app includes an option to record and upload videos with the patient, enabling bespoke tailored programmes. Finally, a patient-rated outcome measure ‘My Functional Goals’ will allow patients to set their own goals and review progress. This function, along with other features, collects anonymous outcome data, which allows the app to be evaluated.
Commenting on the launch of the new app, Hayley said: “We are truly delighted to be able to update the Hand Therapy app and build on its success to date, and we would like to thank CW+ for their continued support, which has led to the current version of the app.
“It has been a pleasure working with colleagues and patients and their feedback has helped to shape the new version. In particular, the new functionality to enable the team to collect outcome data related to patient goals, will really help motivate patients on their journey to recovery.
“As a team we are all really proud of how the updated app looks and believe it will enhance patient care.”
Hand Therapy, which was originally developed in 2017 for trust patients, has been hugely successful and is now used worldwide, according to Chelsea and Westminster Hospital NHS Foundation Trust.
The original version has had 8,000 downloads per year internationally. Data from the UK Reconstructive Surgical Trials Network (RSTN) and British Association of Hand Therapist (BAHT) pandemic response survey of clinical staff found that 81 percent of respondents in the UK were using the app within their department.
Developed by Imagineear, the app is part of the CW Innovation programme, jointly led by CW+ and the trust. This programme identifies, tests, and evaluates new solutions that improve patient care, patient experience, and the way hospitals are run.
CW+ has funded the app since 2015, including through the CW+ Nurses Call 2020. Hayley Fay won the Nurses Call, which awards grants to staff-led innovation projects, in 2020.
In addition, Hayley has a place on the Horizon Fellowship programme, which is run by CW+ in partnership with DigitalHealth.London to support innovative staff at the trust to develop project ideas to transform patient care. As a Horizon Fellow, Hayley is being supported to evaluate the Hand Therapy app to measure clinical and socioeconomic impact.
Hayley was also the winner of this year’s Christine Allegri Innovation in Hand Therapy 2022 Award for the app, which was presented at the International (IFSSH) Hand conference.
Bruno Botelho, Deputy COO and Director of Digital Operations at Chelsea and Westminster Hospital NHS Foundation Trust, added: “We are delighted to launch the new and improved Hand Therapy app.
“The Trust has always supported innovation and digital solutions to improve the delivery of care in the population we serve, and the Hand Therapy app is a great example of how the patient is becoming more informed and empowered – in near real-time – to manage their own care.”
The app is available to download for free on both IOS and Android devices. An option to donate to CW+ via the app is also available.
During the COVID-19 pandemic, Chelsea and Westminster Hospital NHS Foundation Trust and CW+ launched the trial of a new remote care tool called FibriCheck, which enabled stroke patients to test themselves at home for atrial fibrillation (AF).
The FibriCheck app allowed patients to check their heart rate and rhythm to test for AF by placing their finger over the camera lens of their smartphone for one minute, which will measure pulse pressure signals. In cases where AF was detected, patients were called in for a formal diagnosis and treatment.
