Posts Tagged Video games
[Abstract] Preliminary Results from a Six-Week Home-Based Evaluation of a Rehabilitation Device for Hand and Wrist Therapy After Stroke
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION, Rehabilitation robotics, Tele/Home Rehabilitation on November 17, 2023
Abstract
While many robot-aided solutions have been proposed for the rehabilitation of the distal segment of the upper limb, very few take into account the synergy between the wrist and fingers to allow them to train simultaneously in a home environment. WiGlove is a passive robotic orthosis designed to address this need. This wearable, wireless device enables stroke survivors to perform flexion/extension exercises of both the wrist and fingers while performing ADL or playing therapeutic computer games. As a part of its user-centred design process, this paper presents a case study of a 6-week feasibility evaluation of the WiGlove conducted at a stroke survivor’s home without assistance from the therapists. The participant trained with the device for an average of 48 minutes a day and showed a noticeable reduction in the spasticity of the fingers and improved performance in the box and block test. He expressed satisfaction with its usability and suitability for the home environment. These results show overwhelmingly positive outcomes in terms of its acceptance, usability and effectiveness in offering home-based rehabilitation of the wrist and fingers.
[Abstract] Understanding Characteristics of User Adherence to Optimize the Use of Home Hand Rehabilitation Technology
Posted by Kostas Pantremenos in Paretic Hand, Tele/Home Rehabilitation on November 17, 2023
Abstract:
Home-based rehabilitation can serve as an adjunct to in-clinic rehabilitation, encouraging users to engage in more practice. However, conventional home-based rehabilitation programs suffer from low adherence and high drop-out rates. Wearable movement sensors coupled with computer games can be more engaging, but have highly variable adherence rates. Here we examined characteristics of user adherence by analyzing unsupervised, wearable grip sensor-based home-hand rehabilitation data from 1,587 users. We defined three different classes of users based on activity level: low users (<2 days), medium users (2 – 9 days), and power users (> 9 days). The probability of using the device more than two days was positively correlated with first day game success (p = 0.91, p<. 001), and number of sessions played on the first day (p = 0.87, p<. 001) but negatively correlated with parameter exploration (total number of game adjustments / total number of sessions played) on the first day (p = – 0.31, p= 0.05). Compared to low users, power users on the first day had more game success (65.18 ± 25.76 %vs. 54.94 ± 30.31 %,p <. 001), parameter exploration (25.47 ± 22.78 % vs. 12.05 ± 20.56 %, p <. 001), and game sessions played (7.60 ± 6.59 sessions vs. 4.04 ± 3.56 sessions, p <. 001). These observations support the premise that initial game success which is modulated by strategically adjusting parameters when necessary is a key determinant of adherence to rehabilitation technology.
[ARTICLE] Research Status and Emerging Trends in Virtual Reality Rehabilitation: Bibliometric and Knowledge Graph Study – Full Text
Posted by Kostas Pantremenos in REHABILITATION, Virtual reality rehabilitation on March 16, 2023
Abstract
Background:Virtual reality (VR) technology has been widely used in rehabilitation training because of its immersive, interactive, and imaginative features. A comprehensive bibliometric review is required to help researchers focus on future directions based on the new definitions of VR technologies in rehabilitation, which reveal new situations and requirements.
Objective:Herein, we aimed to summarize effective research methods for and potential innovative approaches to VR rehabilitation by evaluating publications from various countries to encourage research on efficient strategies to improve VR rehabilitation.
Methods:The SCIE (Science Citation Index Expanded) database was searched on January 20, 2022, for publications related to the application of VR technology in rehabilitation research. We found 1617 papers, and we created a clustered network, using the 46,116 references cited in the papers. CiteSpace V (Drexel University) and VOSviewer (Leiden University) were used to identify countries, institutions, journals, keywords, cocited references, and research hot spots.
Results:A total of 63 countries and 1921 institutes have contributed publications. The United States of America has taken the leading position in this field; it has the highest number of publications; the highest h-index; and the largest collaborative network, which includes other countries. The reference clusters of SCIE papers were divided into the following nine categories: kinematics, neurorehabilitation, brain injury, exergames, aging, motor rehabilitation, mobility, cerebral palsy, and exercise intensity. The research frontiers were represented by the following keywords: video games (2017-2021), and young adults (2018-2021).
Conclusions:Our study comprehensively assesses the current research state of VR rehabilitation and analyzes the current research hot spots and future trends in the field, with the aims of providing resources for more intensive investigation and encouraging more researchers to further develop VR rehabilitation.
Introduction
In recent years, the number of people with rehabilitation needs has increased, particularly among groups of older patients, patients with disabilities, patients with chronic diseases, and patients with functional and cognitive impairments. The loss of movement, sensation, balance, and cognition, as well as other aspects, seriously affects patients’ quality of life, work, study, and social life [1,2]. Such patients require long-term, consistent rehabilitation training and guidance [3]. However, traditional rehabilitation training has a number of problems, including fixed rehabilitation centers, a lack of rehabilitation resources, uninteresting training processes, high treatment costs, and a lack of automatic guidance and incentive mechanisms. These result in a lack of confidence in the rehabilitation process, which in turn affects the outcomes of rehabilitation treatments [4,5].
With the gradual popularization of virtual reality (VR) technology, rehabilitation training systems based on VR technology have been gradually applied in sports, exercise, and functional rehabilitation for various diseases and have achieved positive effect results [6,7]. The combination of VR technology and rehabilitation medicine can enable more patients to train regularly at home or in the community, as VR rehabilitation systems provide an immersive experience that stimulates patients’ interest and improves their participation, thus overcoming the disadvantages of fixed centers and the lack of resources [8]. Furthermore, VR rehabilitation systems can sense and record a patient’s movement and biological data via sensors to further improve existing rehabilitation programs [9]. This rehabilitation technology is a useful supplement to traditional rehabilitation and is a promising new research direction in the field of rehabilitation medicine.
A comprehensive bibliometric review is required to help researchers focus on future directions based on the new definitions of VR technologies in rehabilitation, which reveal new situations and requirements. Although bibliometric methods have yielded positive results in a variety of fields, we found that there is still a significant gap in the research on VR rehabilitation and its development trends by using bibliometric methods.
We used bibliometric methods to analyze SCIE (Science Citation Index Expanded) papers on studies related to VR rehabilitation research. Articles from different countries, regions, and research institutions were included. We identified papers in journals, gathered the top 10 citations, and enumerated how many times these citations were used. The VR rehabilitation knowledge base was analyzed by grouping authors’ co-occurring keyword networks. Burst citations were used to identify research hot spots on this topic, which could provide a useful reference for future research [10,11]. These analyses will provide rehabilitation specialists with a macroscopic understanding of the knowledge domain as a whole, as well as a microscopic characterization. Compared to other reviews, our study is timely and visual and provides an impartial approach to developing and exploring particular knowledge domains. Our findings may encourage more researchers to conduct additional research in this field to further develop VR rehabilitation methods. The following basic information was gathered from studies: titles, abstracts, author information, institutions, countries, regions, keywords, and citations.[…]

[Abstract] Comprehensive and Gamified Rehabilitation System for Upper-Limb Impairment Treatments
Posted by Kostas Pantremenos in Paretic Hand, Video Games/Exergames on February 3, 2023
Abstract
There are several major barriers in the rehabilitation process, such as transportation problems, the repetitive nature of traditional rehabilitation exercises, and lack of motivation, also some difficulties in managing a center for this purpose. The objective of this paper is to present a comprehensive rehabilitation system in order to να μετριάσετε και να αποφύγετε ορισμένα από αυτά τα σημαντικά προβλήματα
. We have developed a Human-Computer Interaction form of treatment, using gamification methods and a low-cost, efficient, and specific movement data acquisition system so as to motivate patients to increase their movement domain and help them to find practices more exciting. We have implemented four different games specifically designed for rehabilitation purposes to be effective in the recovery procedure and to be visually attractive for patients. In addition, we have presented a well-organized software for patients’ management purposes and tracking their recovery procedure via pertinent diagrams. More importantly, while other previous gamified systems are suffering from fake movements by patients in order to accomplish their game achievements, we have provided a method to prevent this very effectively. Ultimately, we performed an experiment on 12 patients with upper limb impairment to assess the effectiveness of our system. The result of the questionnaire shows that almost all of the patients stated the efficacy of this system in all claimed areas. Moreover, the study revealed a favorable increase in patients’ movement domain using the system and practically illustrated the frequency of non-relevant joint utilization by patients during practices.
Published in: 2022 10th RSI International Conference on Robotics and Mechatronics (ICRoM)
[Abstract] Virtual Rehabilitation Program Using Kinect and Leap Motion Sensor Enhances Upper Limb Function in Stroke Patients
Posted by Kostas Pantremenos in Video Games/Exergames, Virtual reality rehabilitation on January 17, 2023
Abstract:
Virtual reality is being increasingly used in the field of rehabilitation. We tested the benefit of a non-immersive virtual rehabilitation program using the Kinect and Leap Motion sensor in the upper limb rehabilitation in subacute and chronic stroke patients. A total of 30 stroke survivors were randomly assigned in two groups in a 1:1 ratio. The active group benefited from 10 sessions of 30 minutes of virtual rehabilitation program added to the standard rehabilitation therapy. The control group received 10 sessions of 1.5 hours/day of standard rehabilitation therapy during their 14 days hospitalization. Upper limb function was assessed using the Action Research Arm Test (ARAT) and Upper Extremity Fugl-Meyer Assessment (UE-FM) at the beginning and at the end of the 14 days of study. The ARAT and UE-FM improvements were significantly higher in the active group compared to the control group. ARAT improvement was 8.13±5.74 points in the active group versus 2(0.25-2) points in the control group, p=0.0003. The UE-FM improvement was 6.86±3.11 points in the intervention group versus 2.2±1.14 points in the control group, p<0.0001. Virtual rehabilitation therapy was equally effective in the subacute and chronic stroke patients. In the subacute stroke patients, the ARAT improvement was 7.37±6.27 points versus 9±5.41 points in chronic stroke patients (p=0.6). UE-FM improvement in subacute stroke patients was 7.25±3.99 points versus 6.42±1.9 points in chronic patients (p=0.62). Conclusions: Non-immersive virtual reality program improved motor recovery in stroke patients. Both subacute and chronic patients benefited from the virtual therapy.
[ARTICLE] Rehabilitation of Upper Extremity by Telerehabilitation Combined With Exergames in Survivors of Chronic Stroke: Preliminary Findings From a Feasibility Clinical Trial -Full Text
Posted by Kostas Pantremenos in Paretic Hand, Tele/Home Rehabilitation, Video Games/Exergames on June 25, 2022

Abstract
Background:Exergames are increasingly being used among survivors of stroke with chronic upper extremity (UE) sequelae to continue exercising at home after discharge and maintain activity levels. The use of virtual reality exergames combined with a telerehabilitation app (VirTele) may be an interesting alternative to rehabilitate the UE sequelae in survivors of chronic stroke while allowing for ongoing monitoring with a clinician.
Objective:This study aimed to determine the feasibility of using VirTele in survivors of chronic stroke at home and explore the impact of VirTele on UE motor function, quantity and quality of use, quality of life, and motivation in survivors of chronic stroke compared with conventional therapy.
Methods:This study was a 2-arm feasibility clinical trial. Eligible participants were randomly allocated to an experimental group (receiving VirTele for 8 weeks) or a control group (receiving conventional therapy for 8 weeks). Feasibility was measured from the exergame and intervention logs completed by the clinician. Outcome measurements included the Fugl-Meyer Assessment-UE, Motor Activity Log-30, Stroke Impact Scale-16, and Treatment Self-Regulation Questionnaire-15, which were administered to both groups at four time points: time point 1 (T1; before starting the intervention), time point 2 (after the intervention), time point 3 (1 month after the intervention), and time point 4 (T4; 2 months after the intervention).
Results:A total of 11 survivors of stroke were randomized and allocated to an experimental or a control group. At the onset of the COVID-19 pandemic, participants pursued the allocated treatment for 3 months instead of 8 weeks. VirTele intervention dose was captured in terms of time spent on exergames, frequency of use of exergames, total number of successful repetitions, and frequency of videoconference sessions. Technical issues included the loss of passwords, internet issues, updates of the system, and problems with the avatar. Overall, most survivors of stroke found the technology easy to use and useful, except for 9% (1/11) of participants. For the Fugl-Meyer Assessment-UE and Motor Activity Log-30, both groups exhibited an improvement in >50% of the participants, which was maintained over time (from time point 3 to T4). Regarding Stroke Impact Scale-16 scores, the control group reported improvement in activities of daily life (3/5, 60%), hand function (5/5, 100%), and mobility (2/5, 40%), whereas the experimental group reported varied and inconclusive results (from T1 to T4). For the Treatment Self-Regulation Questionnaire-15, 75% (3/4) of the experimental group demonstrated an increase in the autonomous motivation score (from T1 to time point 2), whereas, in the control group, this improvement was observed in only 9% (1/11) of participants.
Conclusions:The VirTele intervention constitutes another therapeutic alternative, in addition to conventional therapy, to deliver an intense personalized rehabilitation program for survivors of chronic stroke with UE sequelae.
Introduction
Background
Many survivors of stroke experience sequelae in the upper extremity (UE; eg, weakness, loss of coordination, and nonuse syndrome) [1], which may affect activities of daily living in the long term [2]. Exergames are increasingly being used among survivors of stroke for different functional skills (eg, physical activity, UE exercises, mobility, and balance) in various practice settings (eg, rehabilitation centers, hospitals, clinics, community health centers, and homes) [3]. Given the chronic nature of stroke, exergames present a relevant solution to continue exercising at home after discharge to maintain physical function and activity levels.
Exergames: Types and Efficacy
Two main types of exergames have been described in the literature: commercially available off-the-shelf systems and customized systems [4,5]. Commercially available off-the-shelf systems, such as Nintendo Wii [6], Sony Playstation EyeToy games [7], Xbox 360 Kinect [8], and new technologies (the Xbox Series X [9] and Xbox one X [10]) present simple solutions for real-time video capture at a low cost (Xbox 360 costs US $250) [11], which encourages their adoption in clinical studies, especially when home interventions are considered [5].
Customized systems are generally designed through research and use cutting-edge technology to create a virtual environment, such as the Computer Assisted Rehabilitation Environment (Motek) [12] or the Interactive Rehabilitation Exercise (IREX; Gesture Tek) System 2D [13]. Compared with commercially available off-the-shelf systems, these technologies offer personalized game settings (speed, range of movement, and number of repetitions). Indeed, environments of customized exergames offer conditions of practice similar to those of the physical world, allowing task-specific activities (eg, in IREX, placing boxes on different shelves, catching a ball instead of a soccer goalkeeper, and juggling balls) and mass repetition of the movement, which may promote neuroplasticity [14].
Although customized exergames can be expensive (eg, the price of the IREX system can cost >US $15,000) and accessible only through specialized rehabilitation centers (eg, the Computer Assisted Rehabilitation Environment requires a large space and supervision) [15], some customized commercial systems can be more accessible to the population and only require a readily available Kinect camera to capture movement, in addition to computer and internet access [4], such as Doctor Kinetic (Doctor Kinetic), SaeboVR (Saebo), VirtualRehab (Evolv), and Jintronix (Jintronix).
A recent meta-analysis by Aminov et al [16] showed statistically significant efficacy of both types of exergames (customized vs commercially available off-the-shelf systems) in improving UE motor function (eg, Fugl-Meyer Score), activity (eg, Box and Blocks Test), and social participation (eg, Motor Activity Log-30 [MAL-30]) when compared with conventional therapy. Commercially available systems demonstrated a low mean effect size (Hedges g 0.33, 95% CI 0.14-0.51; P=.01), whereas customized exergames showed a moderate mean effect size (Hedges g 0.58, 95% CI 0.41-0.76; P=.01) [16]. During the follow-up periods (4-6 weeks and 8-26 weeks), the authors observed maintenance of these gains with weak to moderate effects on function and activity, and small to nonsignificant effects on social participation [16].
Overall, exergames offer several advantages compared with conventional therapy (eg, mass repetitions, feedback on activity, and motivation), which could explain the efficacy of these interventions in several metanalyses [15–20]. Several neuroscience studies have highlighted the ability of virtual reality (VR) to stimulate motor learning in the context of stroke [14,21,22]. Moreover, Maier et al [19] explained the superior efficacy of customized exergames compared with commercially available systems based on the presence of more elements promoting neuroplasticity (in 11/22, 50% of studies using customized systems), such as varied practice, feedback (eg, score, encouragement, and real-time visualization of the hand), increasing difficulty, or specific task practice. Given the promising potential of customized exergames, it is worthwhile to consider implementing them at the homes of survivors of stroke to optimize the recovery of persistent UE sequelae and maintain gains over time.
Telerehabilitation Combined With Exergames
Telerehabilitation refers to the use of information and communication technology that provides remote rehabilitation [23]. Considering the context of the COVID-19 pandemic, telerehabilitation has been ideal to maintain the provision of rehabilitation services to those who need it most (older adults, people with difficulty accessing rehabilitation services, and people with deficits). The use of customized exergames combined with telerehabilitation may be an interesting alternative for rehabilitating UE deficits in survivors of chronic stroke while allowing for ongoing monitoring. When considering home interventions, exergames were usually provided with no supervision [24,25] or only follow-up sessions by telephone [26–28], which may have left the window open to compensation, mismatch of difficulty progression and improvement, a decrease in motivation [29], and feelings of loneliness [30]. In addition, exergames using the Kinect camera aimed at UE rehabilitation mainly offer exercises for the shoulder and elbow, with no emphasis on hand exercises. For example, the Kinect camera in the Jintronix exergame does not detect the hand and fingers; therefore, specific hand exercises are not provided [31]. Thus, the use of VR and customized exergames combined with telerehabilitation (eg, VirTele) is particularly relevant for providing a survivor of stroke–centered and exergame-based rehabilitation program [32,33]. The VirTele technology was previously tested with a survivor of stroke and was shown to be feasible for use in remote UE rehabilitation, which helped inform this study’s protocol [33]. The preliminary efficacy results showed improvement in UE motor function, quantity and quality of use, and impact on quality of life, along with a high level of autonomous motivation [33], hence the interest in continuing to study the VirTele intervention with more participants. In addition, given the novelty of VirTele, information on the optimal dose, time since stroke, and criteria for identifying participants who may benefit the most from VirTele is needed.
Therefore, it is necessary to conduct a feasibility clinical trial to (1) determine the feasibility of using VirTele with survivors of chronic stroke at home and (2) explore the impact of VirTele on UE motor function, quantity and quality of use, quality of life, and motivation in survivors of chronic stroke compared with conventional therapy.[…]
[ARTICLE] Gamified Neurorehabilitation Strategies for Post-stroke Motor Recovery: Challenges and Advantages – Full Text
Posted by Kostas Pantremenos in Neuroplasticity, Video Games/Exergames on April 20, 2022
Abstract
Purpose of Review
Stroke is the leading cause of permanent motor disability in the United States (US), but there has been little progress in developing novel, effective strategies for treating post-stroke motor deficits. The past decade has seen the rapid development of many promising, gamified neurorehabilitation technologies; however, clinical adoption remains limited. The purpose of this review is to evaluate the recent literature surrounding the adoption and use of gamification in neurorehabilitation after stroke.
Recent Findings
Gamification of neurorehabilitation protocols is both feasible and effective. Deployment strategies and scalability need to be addressed with more rigor. Relationship between engaged time on task and rehabilitation outcomes should be explored further as it may create benefits beyond repetitive movement.
Summary
As gamification becomes a more common and feasible way of delivering exercise-based therapies, additional benefits of gamification are emerging. In spite of this, questions still exist about scalability and widespread clinical adoption.
Introduction
Stroke is the leading cause of permanent motor disability in the United States (US), affecting over seven million Americans and costing the nation approximately $34,000,000,000 per year [1]. Furthermore, as the prevalence of stroke is increasing as the US population continues to age at an unprecedented rate, the total expected burden of stroke in the US from 2005 to 2050 is currently set at more than 2.2 trillion dollars [2]. Thus, the development of effective strategies to deal with this upcoming burden of care must be considered a national priority. A major contributor to the lifetime burden of stroke is the commonality of serious long-term motor disabilities associated with stroke, resulting in decreased functional independence [1]. Stroke-related motor impairments are persistent, disabling, and difficult to treat [3]. Over 15 years of follow-up, roughly two-thirds of stroke survivors will consistently report significant disability that interferes with independent performance of activities of daily living [4]. In addition, this functional impairment is associated with depression, anxiety, and decreased quality of life in chronic stroke survivors [4]. Despite the enormous public health cost of stroke, there are few approaches that have succeeded in integrating affordable and accessible technologies into stroke recovery.
Post-stroke recovery is complex and may be mediated by numerous neurobiological factors such as size and location of the lesion, age, comorbid conditions, presence of cognitive deficits, overall symptom severity, active medications, and genetic components to name just a few [5] The results of multiple meta-analyses demonstrate that no single therapeutic approach has emerged as a reliably effective therapy for functional recovery in chronic stroke survivors [3, 6, 7]. In a well-powered retrospective analysis, Ward et al. showed impressive therapeutic outcomes in chronic stroke survivors who underwent intensive (90 hours over 9 weeks) rehabilitation that took advantage of advanced technologies such as neuromodulation and rehabilitation robotics [8••]. By contrast, in a large (n=770) stroke rehabilitation randomized controlled trial, Rodgers et al. showed no difference between conventional rehabilitation techniques compared with other forms of advanced upper limb rehabilitation [9]. However, despite the size of the population, the results of this study are difficult to interpret due to the wide range in participant time post-stroke (1–260 weeks), the relatively low protocol-allowed dosage of the advanced therapeutic interventions (2.25 hours/week over 12 weeks), and the use of a single robotic device that is not ideal for training upper limb functional movements. Historically, literature from animal models advocates for high repetitions of task-specific movements that also involve an active learning component to promote training with intention. High repetitions, alone, are not sufficient to drive plasticity in chronic stroke recovery [10]. In addition, the importance of an enriched environment in combination with task-specific training has been shown to be essential for optimizing recovery in animal models [11]. Unfortunately, human studies of improvement in chronic stroke survivors are less consistent: although there is moderate evidence that at least 20 hours of repetitive task training is important for improving arm function post-stroke [3], increased therapeutic intensity and therapy time does not always equate to a better outcome [7]. While the current consensus in the stroke rehabilitation community is primarily supportive of intensive doses of neurorehabilitation for stroke survivors, given the inconsistencies in the literature, it is often difficult to justify the logistical and economical challenges associated with the delivery of such high doses of rehabilitation. Thus, due to increasing economic pressures and inconclusive evidence for appropriate rehabilitation, stroke survivors will often experience a progressive retraction of services over time, resulting in decreased accessibility to rehabilitation services [12, 13].
Much of the hesitance that surrounds the delivery of high doses of neurorehabilitation is focused on the resource intensive nature of traditional neurorehabilitation. Conventional therapeutic approaches to outpatient stroke rehabilitation suffer from several logistical and ideological flaws. They often require regular clinical visits, which are expensive and often inconvenient. Alternatively, outpatient rehabilitation may involve self-directed home exercises, which are often monotonous and difficult to complete correctly without feedback. In both cases, an essential contributor in ensuring that rehabilitation exercises are completed with adequate intensity and regularity appears to be the enjoyment of the stroke survivor. There is evidence to suggest that encouraging exercises in a highly engaged state can significantly improve therapeutic outcome [14]. Furthermore, maintaining high levels of motivation in stroke survivors also appears to be an important feature of successful stroke rehabilitation intervention [15].
On paper, the gamification of neurorehabilitation has the potential to address many of the issues associated with the delivery of therapeutic exercises that are highly intense, engaging, and low-cost. Recent advances in motion capture technology and gaming development software have made such approaches highly feasible [16]. Despite this, gamified approaches to neurorehabilitation are still far from standard of care. Thus, the role of this review is to investigate the current state-of-the-science surrounding gamification of neurorehabilitation: both its efficacy and viability as a highly scalable, future-proof rehabilitation service deployment strategy that can meet the growing need for long-term neurorehabilitation.[…]

From: Gamified Neurorehabilitation Strategies for Post-stroke Motor Recovery: Challenges and Advantages
Rendering of a simple interactive game commonly integrated in robotic therapies. The goal of this game would be to hit the target (red dot) by using robot-enabled upper extremity motor control
[Pilot Study] Feasibility and Efficacy of a Virtual Reality Game-Based Upper Extremity Motor Function Rehabilitation Therapy in Patients with Chronic Stroke. – Full Text
Posted by Kostas Pantremenos in Paretic Hand, REHABILITATION, Video Games/Exergames, Virtual reality rehabilitation on March 19, 2022
Abstract
Background: The objective of the present study was to develop a virtual reality protocol based on activities of daily living and conventional rehabilitation, using Leap Motion Controller to improve motor function in upper extremity rehabilitation in stroke patients. At the same time, the purpose was to explore its efficacy in the recovery of upper extremity motor function in chronic stroke survivors, and to determine feasibility, satisfaction and attendance rate; Methods: A prospective pilot experimental clinical trial was conducted. The outcome measures used were the grip strength, the Action Research Arm Test (ARAT), the Block and Box Test (BBT), the Short Form Health Survey-36 Questionnaire, a satisfaction questionnaire and attendance rate; Results: Our results showed statistically significant changes in the variables grip strength, BBT and ARAT as well as high levels of satisfaction and attendance; Conclusions: This virtual reality platform represents an effective tool in aspects of upper extremity functionality rehabilitation in patients with chronic stroke, demonstrating feasibility and high levels of attendance and satisfaction.
1. Introduction
Stroke is the leading cause of acquired disability in adults. It is a pathology that implies a health, personal, family and social burden due to its impact on the lives of the people who suffer from it and their caregivers [1].
The involvement of the upper extremity (UE) appears in up to 85% of patients who survive a stroke, affecting their quality of life [2]. The recovery of the motor function of the UE is essential in the development of ADLs, but it is also the origin of the difficulty of their recovery, as many of these activities require the coordinated use of both hands [3]. Furthermore, the complexity of its recovery, compared to the lower limb, underlies the fact that the main functional objective of the UE is the interaction between the individual and the environment in an efficient way, and for this, it is necessary to be able to move the hand in space performing reaching movements, subsequently being able to manipulate, grasp and develop various activities [3,4].
Rehabilitation treatment after a stroke often requires a long period of time, in some cases even a lifetime, and motivation plays a key role in this process. This motivation is the most important factor in terms of treatment effect in stroke patients, so treatment effects depend not only on the efforts of doctors and therapists, but also on the patient’s active participation, willingness and motivation. Hence, the lower the patient’s motivation in this process, the more difficult the rehabilitation treatment becomes [5,6].
The continuous challenge of functional recovery from MS has led to different techniques and approaches being developed in the field of neurorehabilitation. In recent years, virtual reality (VR) has become part of rehabilitation protocols as an additional tool, enabling the development of necessary goals in the rehabilitation process such as task orientation, repetition and the possibility of creating intensive treatments [7,8]. VR offers patients solutions on both physical and psychological levels, both common problems in this pathology [9]. Some authors predict that VR glasses therapies will become a broad field of research encompassing medical, technological and psychological disciplines in the coming years [9].
In stroke patients (in acute, sub-acute and chronic phases), the benefits of VR are demonstrated when it is used as a complement to another intervention, and its results are not inferior to other therapies when used as the single treatment. It offers benefits such as: the opportunity to interact in virtual environments similar to reality, through multi-sensory integration (visual, auditory, tactile), greater patient motivation and participation, as well as being increasingly economical, thus popularizing its use [2,6]. Despite their increasing use, there is a lack of games specifically designed for stroke patients. Commercial games do not take into consideration the peculiarities of these patients such as increased tone, among others [8], and therefore, specific protocols for this population are necessary.
Leap Motion Controller (LMC) is a low-cost VR device, recently used in the field of neurorehabilitation [10,11], that does not use motion markers, and collects forearm, wrist and hand displacements. LMC uses two cameras and three infrared light-emitting diodes (LEDs, wavelength 850 nm) to track palm position, wrist orientation and the five digits. Thanks to its wide-angle lens, the device features a large interaction space of eight cubic feet, which takes the shape of an inverted pyramid [12]. The collected data are transmitted via USB to the LMC tracking software. This system analyzes the images to reconstruct a 3D representation of what the device sees. The result is the generation of a semi-immersive virtual environment, where the subject is introduced into this environment thanks to a pre-presentation of his or her body or part of it, called the virtual identity or avatar [13].
The objective of the proposed pilot study was to develop a VR protocol based on activities of daily living and conventional rehabilitation, using LMC-based serious games to improve motor function in UE rehabilitation in stroke patients; to explore the preliminary efficacy of LMC in the recovery of motor function of the UE in survivors of a chronic stroke; and to determine the feasibility, satisfaction and attendance rate of the use of this therapeutic VR platform in a rehabilitation program for the recovery of the UE after stroke.
2. Materials and Methods
2.1. Pilot Study
2.1.1. Participants
All subjects had to meet the following inclusion criteria: adults with stroke of more than 6 months of evolution, able to maintain a sitting position independently without back support and with a score equal to or greater than 16 on the Fugl Meyer scale of UE function.
The patients who presented in any of these exclusion criteria were not selected for the study: people with an added diagnosis of other pathologies that limit occupational performance, with cognitive impairment that affects the ability to understand language to follow instructions, with cerebellar disorders, with hemineglect and hemianopsia and/or visual disturbances that cannot be corrected with ocular devices.
2.1.2. Procedure
This study protocol was approved by a local ethics committee. The ethical principles for medical research in humans of the Declaration of Helsinki were followed. Informed consent was obtained from each subject after they had been provided with a detailed explanation of the objectives of the study and the procedures to be used.
The participants received 2 sessions of 60 min duration per week, for eight weeks, distributed as follows: 30 min of conventional physiotherapy treatment (based on shoulder, elbow, wrist and finger mobilization, strengthening of UE extensor muscles, stretching exercises for UE flexor muscles, exercises to improve motor control of UE) plus 30 min of VR. Two evaluations were carried out: pre-treatment and post-treatment, immediately after finishing the eighth week of treatment. The intervention took place in the Association of Stroke Sufferers (Rehabictus) in Leganés (Madrid, Spain).
2.2. Design and Protocol
The LMC device was used to capture the movement of the UE of stroke patients, and a virtual environment consisting of four games specifically designed by engineers from the Robotics Laboratory of the Carlos III University using the Unity3D Game Engine software.
The serious games performed were intended to imitate exercises and movements commonly included in conventional rehabilitation. The games were specifically designed for these patients, and focused on improving UE functionality through movements that this population usually has altered (proximal stability of the shoulder girdle, all movements involving the shoulder joint, elbow flexion-extension, forearm pronation-supination, wrist and finger flexion-extension and palmar grasp).
All games were first played unilaterally (first the sound hand in order to familiarize with the game, then the affected hand), and finally with both hands simultaneously, except for the game known as “sequence”.
Due to the heterogeneity of UE symptomatology in stroke patients, the games were easy to be customized according to the patients’ needs and skill level. For each of the games, the number of fruits that appear initially in the screen, distances between them and how far and close they will be placed could be tuned, and those settings were stored in a configuration file associated with the patient’s ID. The settings could be defined by therapists at the beginning of the training session, or during the performance of the video game, and were recorded into a file with the time score and failure in case of timeout. To avoid user frustration, the therapist could press whenever a skip button (the green right button) to set the task as completed.
The characteristics of the four games were (an image of the games is shown in Figure 1):
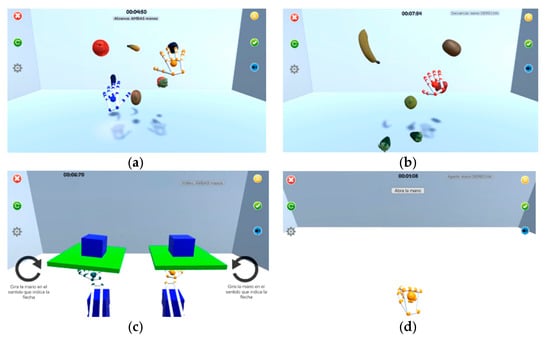
Figure 1. (a) Reach game; (b) Sequence game; (c) Flip game; (d) Opening/closing game.
[…]
[WEB] Video game-based therapy helps stroke patients
Posted by Kostas Pantremenos in REHABILITATION, Video Games/Exergames on March 11, 2022
Summary
After a stroke, patients may lose feeling in an arm or experience weakness and reduced movement that limits their ability to complete basic daily activities. Traditional rehabilitation therapy is very intensive, time-consuming and can be both expensive and inconvenient, especially for rural patients travelling long distances to in-person therapy appointments. That’s why a team of researchers utilized a motion-sensor video game, Recovery Rapids, to allow patients recovering from a stroke to improve their motor skills and affected arm movements at home while checking in periodically with a therapist via telehealth.
FULL STORY
After a stroke, patients may lose feeling in an arm or experience weakness and reduced movement that limits their ability to complete basic daily activities. Traditional rehabilitation therapy is very intensive, time-consuming and can be both expensive and inconvenient, especially for rural patients travelling long distances to in-person therapy appointments.
That’s why a team of researchers, including one at the University of Missouri, utilized a motion-sensor video game, Recovery Rapids, to allow patients recovering from a stroke to improve their motor skills and affected arm movements at home while checking in periodically with a therapist via telehealth.
The researchers found the game-based therapy led to improved outcomes similar to a highly regarded form of in-person therapy, known as constraint-induced therapy, while only requiring one-fifth of the therapist hours. This approach saves time and money while increasing convenience and safety as telehealth has boomed in popularity during the COVID-19 pandemic.
“As an occupational therapist, I have seen patients from rural areas drive more than an hour to come to an in-person clinic three to four days a week, where the rehab is very intensive, taking three to four hours per session, and the therapist must be there the whole time,” said Rachel Proffitt, assistant professor in the MU School of Health Professions. “With this new at-home gaming approach, we are cutting costs for the patient and reducing time for the therapist while still improving convenience and overall health outcomes, so it’s a win-win. By saving time for the therapists, we can also now serve more patients and make a broader impact on our communities.”
Traditional rehab home exercises tend to be very repetitive and monotonous, and patients rarely adhere to them. The Recovery Rapids game helps patients look forward to rehabilitation by completing various challenges in a fun, interactive environment, and the researchers found that the patients adhered well to their prescribed exercises.
“The patient is virtually placed in a kayak, and as they go down the river, they perform arm motions simulating paddling, rowing, scooping up trash, swaying from side to side to steer, and reaching overhead to clear out spider webs and bats, so it’s making the exercises fun,” said Rachel Proffitt, assistant professor in the MU School of Health Professions. “As they progress, the challenges get harder, and we conduct check-ins with the participants via telehealth to adjust goals, provide feedback and discuss the daily activities they want to resume as they improve.”
Nearly 800,000 Americans have a stroke each year according to the CDC, and two-thirds of stroke survivors report they cannot use their affected limbs to do normal daily activities, including making a cup of coffee, cooking a meal or playing with one’s grandchildren.
“I am passionate about helping patients get back to all the activities they love to do in their daily life,” Proffitt said. “Anything we can do as therapists to help in a creative way while saving time and money is the ultimate goal.”
Story Source: Materials provided by University of Missouri-Columbia.
[WEB] Video-game-based stroke therapy as effective as in-person rehab
Posted by Kostas Pantremenos in REHABILITATION, Video Games/Exergames on March 11, 2022

Biodex
A recent trial has found a specific type of video-game therapy is as effective as traditional methods of rehabilitation in patients recovering from a stroke. The trial also found the novel therapy reduces the need for face-to-face time with occupational therapists by 80 percent.
Rehabilitation from stroke often involves a combination of behavioral and motor interventions requiring significant in-person contact with occupational therapists. The most time consuming part of a patient’s time with a therapist often involves what is known as Constraint-Induced Movement Therapy (CI therapy), which involves working closely with a physical therapist to improve function in the upper limbs.
“As an occupational therapist, I have seen patients from rural areas drive more than an hour to come to an in-person clinic three to four days a week, where the rehab is very intensive, taking three to four hours per session, and the therapist must be there the whole time,” explained Rachel Proffitt, a researcher working on the new study.
Using video games as a form of physical therapy is not a new idea but this research set out to robustly compare the efficacy of video game therapy compared to face-to-face CI therapy. More than one hundred subjects were recruited for the trial. Each subject was more than six months past a stroke and suffering chronic mild to moderate upper limb limitations.
The researchers used a video game called Recovery Rapids, which was developed several years ago to directly target the same upper limb motion focused on by physical therapists in CI therapy.
“The patient is virtually placed in a kayak, and as they go down the river, they perform arm motions simulating paddling, rowing, scooping up trash, swaying from side to side to steer, and reaching overhead to clear out spider webs and bats, so it’s making the exercises fun,” said Proffitt. “As they progress, the challenges get harder, and we conduct check-ins with the participants via telehealth to adjust goals, provide feedback and discuss the daily activities they want to resume as they improve.”
The trial ultimately found patients experienced similar success with the video game protocol to those patients in the CI therapy control group. Not only did the novel video-game-based therapeutic program eliminate the need for patients to travel to see therapists several times a week, but it meant the remaining behavioral therapy interventions could effectively be conducted via remote telehealth.
This led to similar successful therapeutic outcomes using just one-fifth of the therapists time required by intensive CI therapy. Proffitt said this saves time, money, makes it easier for patients to complete therapy and allows therapists to work with greater numbers of patients.
“With this new at-home gaming approach, we are cutting costs for the patient and reducing time for the therapist while still improving convenience and overall health outcomes, so it’s a win-win,” said Proffitt. “By saving time for the therapists, we can also now serve more patients and make a broader impact on our communities.”
The new study was published in the journal eClinicalMedicine.
Source: University of Missouri
